ABSTRACT
Objective: To evaluate the efficacy of a mindfulness-based treatment (MBT) for smoking cessation or reduction and compare it with that of cognitive behavioral therapy (CBT). Methods: This was a single-center randomized controlled clinical trial including 113 patients divided into two groups: MBT (n = 54) and CBT (n = 59). The interventions comprised eight 90-min sessions. The primary outcome was smoking cessation at 16 weeks after program initiation. Secondary outcomes included reduction in the mean number of cigarettes smoked/day at 16 weeks after treatment initiation, as well as smoking cessation and reduction in the number of cigarettes smoked/day at the last program session. Participants had to attend = 50% of the sessions to be included in the primary outcome analysis. An intention-to-treat analysis was also performed. Results: There was no difference between the groups regarding the primary outcome (30.4% in the MBT group vs. 31.6% in the CBT group, p = 0.68) or immediate abstinence rates (47.8% in the MBT group vs. 36.8% in the CBT group, p = 0.47). Both treatments were equally effective in reducing the number of cigarettes smoked/day at the last program session (a reduction of 93.33% [0-100%] in the MBT group and of 70% [33.3-100%] in the CBT group, p = 0.92) and at 16 weeks after program initiation (a reduction of 57.1% [0-100%] in the MBT group and of 70% [25-100%] in the CBT group, p = 0.49). Conclusions: MBT appears to be as effective as CBT for smoking cessation or reduction and can be an option for the treatment of tobacco use disorders in Brazil
(Brazilian Registry of Clinical Trials identifier: RBR-3w2scz [http://www.ensaiosclinicos.gov.br])
Keywords:
Mindfulness; Smoking cessation; Tobacco use disorder; Psychotherapy, group; Meditation; Cognitive behavioral therapy.
RESUMO
Objetivo: Avaliar a eficácia de um mindfulness treatment (MT, tratamento baseado em atenção plena) para a cessação ou redução do tabagismo e compará-la à da terapia cognitivo-comportamental (TCC). Métodos: Ensaio clínico controlado randomizado realizado em um único centro, com 113 pacientes divididos em dois grupos: MT (n = 54) e TCC (n = 59). As intervenções consistiram em oito sessões de 90 min cada. O desfecho primário foi a cessação do tabagismo 16 semanas após o início do programa. Os desfechos secundários foram a redução da média de cigarros fumados/dia em 16 semanas após o início do programa, bem como a cessação do tabagismo e redução do número de cigarros fumados/dia na última sessão do programa. Os participantes deveriam comparecer a ≥ 50% das sessões para que fossem incluídos na análise do desfecho primário. Foi também realizada uma análise por intenção de tratamento. Resultados: Não houve diferença entre os grupos quanto ao desfecho primário (30,4% no grupo MT vs. 31,6% no grupo TCC, p = 0,68) ou às taxas de abstinência imediata (47,8% no grupo MT vs. 36,8% no grupo TCC, p = 0,47). Ambos os tratamentos foram igualmente eficazes na redução do número de cigarros fumados/dia na última sessão do programa [redução de 93,33% (0-100%) no grupo MT e de 70% (33,3-100%) no grupo TCC, p = 0,92] e em 16 semanas após o início do programa [redução de 57,1% (0-100%) no grupo MT e de 70% (25-100%) no grupo TCC, p = 0,49]. Conclusões: A MT parece ser tão eficaz quanto a TCC para a cessação ou redução do tabagismo e pode ser uma opção para o tratamento do tabagismo no Brasil.
[Registro Brasileiro de Ensaios Clínicos – ReBEC; número de identificação: RBR-3w2scz (http://www.ensaiosclinicos.gov.br)]
Palavras-chave:
Atenção plena; Abandono do hábito de fumar; Tabagismo; Psicoterapia de grupo; Meditação; Terapia cognitivo-comportamental.
INTRODUÇÃO O tabagismo é a principal causa de morte evitável no mundo, com aproximadamente 8 milhões de mortes anuais.(1) No Brasil, 9,8% dos adultos são atualmente fumantes,(2) e estima-se que 428 pessoas morrem todos os dias em virtude de doenças relacionadas ao tabagismo. (3) Além disso, o tabagismo custa ao país 56,9 bilhões de reais em assistência à saúde e perda de produtividade. (3) Embora a maioria dos fumantes afirme que deseja parar de fumar, apenas 1-5% são capazes de deixar de fumar por um longo tempo sem ajuda profissional.(4,5)
A maioria dos tratamentos disponíveis atualmente no Brasil baseia-se na terapia cognitivo-comportamental (TCC), incluindo o tratamento para cessação do tabagismo utilizado no Sistema Único de Saúde. Farmacoterapias podem ser usadas em conjunto com a TCC, principalmente quando o grau de dependência de nicotina é alto. Embora essa estratégia tenha produzido resultados positivos em curto prazo, as taxas de cessação tendem a diminuir significativamente em longo prazo.(6)
A atenção plena (mindfulness) é a habilidade de prestar atenção ao momento presente, intencionalmente e sem julgamentos.(7) Mindfulness treatments (MT, tratamentos baseados em atenção plena) foram originalmente usados para combater transtornos de estresse e dor crônica.(8) Posteriormente, foram usados com sucesso no tratamento de depressão, ansiedade,(9) transtornos alimentares e vícios,(10) e só recentemente foram adaptados para a cessação do tabagismo.(11)
O MT voltado para a cessação do tabagismo usa técnicas de meditação para aumentar a consciência (awareness) de pensamentos, sentimentos e sensações, especialmente aqueles relacionados ao forte desejo de fumar (fissura). O objetivo é ensinar os participantes a sair do “piloto automático” que leva ao tabagismo e a lidar com o desejo de fumar até que ele não mais ocorra. Diferentemente do que ocorre na TCC, o foco não está em evitar gatilhos, e não são usados substitutos.(11)
A eficácia do MT para a cessação do tabagismo mostrou-se semelhante à da TCC ou até mesmo maior no que tange à manutenção da abstinência em longo prazo.(11-13) Além disso, demonstrou-se que o MT está relacionado com melhora do humor, redução do desejo de fumar e redução dos sintomas de abstinência.(14-16)
Pelo que sabemos, nenhum estudo no Brasil avaliou MT para a cessação do tabagismo. O objetivo do presente estudo foi avaliar a eficácia de um MT para a redução ou cessação do tabagismo e compará-la à da TCC.
MÉTODOS Trata-se de um ensaio clínico controlado randomizado aberto realizado entre maio de 2019 e janeiro de 2020 em um único centro, o Hospital de Clínicas da Universidade Federal do Paraná, em Curitiba (PR). O estudo foi aprovado pelo comitê de ética em pesquisa da instituição (Protocolo n. CAAE 02984118.8.0000.0096) e registrado no Registro Brasileiro de Ensaios Clínicos [ReBEC; número de identificação: RBR-3w2scz (http://www.ensaiosclinicos.gov.br)]. Todos os participantes assinaram um termo de consentimento livre e esclarecido antes da inclusão no estudo.
O cálculo do tamanho da amostra foi realizado com base nos resultados de um estudo anterior.(11) Procuramos incluir um total de 120 participantes. Como ambas as intervenções exigiam participação ativa nas sessões, decidimos dividir os participantes em três grupos para cada intervenção, de forma a manter os grupos suficientemente pequenos (20 participantes por grupo). Os participantes foram recrutados por meio de anúncios na televisão, rádio, Internet e jornais, bem como pôsteres e folhetos oferecendo tratamento não farmacológico para a cessação do tabagismo.
Os critérios de elegibilidade foram os seguintes: idade ≥ 18 anos; fumante com média de consumo de pelo menos 5 cigarros por dia e menos de 3 meses de abstinência no último ano; motivado(a) para deixar de fumar nos próximos 30 dias. Os critérios de exclusão foram os seguintes: dependência ativa de substâncias, inclusive dependência de álcool; esquizofrenia ou transtorno de pânico, diagnosticados em conformidade com os critérios do Manual Diagnóstico e Estatístico de Transtornos Mentais, Quinta Edição(17); analfabetismo; uso de medicamentos para a cessação do tabagismo, incluindo bupropiona, vareniclina e nicotina (independentemente da forma de administração).
Os participantes foram aleatoriamente divididos em intervenção (MT) e controle (TCC) após a estratificação baseada na idade (≥ vs. < 40 anos), sexo, cigarros fumados/dia (> vs. ≤ 20), escolaridade (≥ ensino médio completo ou < ensino médio completo) e pontuação no Teste de Fagerström para Dependência de Nicotina (TFDN) (≥ 6 vs. < 6).
Ambas as intervenções consistiram em sessões de 90 min cada, duas vezes por semana, durante quatro semanas. As sessões de MT basearam-se em um protocolo usado em um estudo anterior.(11) As sessões abordaram conceitos teóricos como aprendizagem associativa, “piloto automático” e a compreensão de pensamentos assim como o são (isto é, pensamentos, e não verdades absolutas). Foram usadas meditações guiadas comuns a diversos programas de atenção plena: body scan, meditação de compaixão ou bondade amorosa (loving-kindness meditation), meditação sentada e atenção plena nas atividades diárias. Também foi ensinada uma técnica para lidar com o desejo de fumar “conscientemente”: a técnica RAIN, do inglês Recognize, Accept, Investigate, and Note what cravings feel like as they arise, isto é, reconhecer, aceitar, investigar e notar como é o desejo de fumar quando surge. As sessões foram conduzidas por um instrutor de atenção plena formado pelo Centro Mente Aberta da Universidade Federal de São Paulo, na cidade de São Paulo (SP), um pneumologista com experiência em cessação do tabagismo e um aluno do curso de graduação em medicina. A TCC baseou-se no programa de cessação do tabagismo do Sistema Único de Saúde,(18) incluindo também exercícios da cartilha “Passos para uma Vida Livre do Tabaco”(19) e do programa Freedom from Smoking da American Lung Association.(20) As sessões foram conduzidas por pneumologistas com experiência em TCC para a cessação do tabagismo e um aluno do curso de graduação em medicina.
Além das sessões presenciais em grupo, sugerimos aos participantes que praticassem em casa e fornecemos materiais extras, tais como panfletos, folhetos e gravações. Incentivamos a redução gradual do consumo de cigarros, e o dia da cessação foi agendado para a quarta sessão em ambos os grupos.
O desfecho primário foi a cessação do tabagismo 16 semanas após o início do programa. Os desfechos secundários foram a redução da média de cigarros fumados/dia em 16 semanas após o início do programa; cessação do tabagismo e redução da média de cigarros fumados/dia 4 semanas após o início do programa (após a última sessão, isto é, cessação imediata) e correlação entre as taxas de comparecimento e a porcentagem de redução da média de cigarros fumados/dia.
Os desfechos foram avaliados por meio de questionários padronizados administrados por entrevistadores que desconheciam a intervenção. A abstinência foi confirmada pela concentração de monóxido de carbono no ar expirado < 8 ppm.(21)
Apenas os participantes que compareceram a no mínimo 50% das sessões foram incluídos na análise primária dos desfechos, pois o processo de aprendizagem exigia uma frequência mínima. Foi realizada uma análise por intenção de tratamento, incluindo todos os participantes que compareceram pelo menos à primeira sessão, bem como uma análise na qual foram incluídos apenas aqueles que compareceram a 75% das sessões. Desistências ocorridas depois da randomização, porém antes da distribuição dos grupos, foram excluídas.
Participantes que perderam o seguimento foram considerados como não abstinentes. Como de praxe em ensaios que examinam a cessação do tabagismo, uma análise last observation carried forward foi realizada para quantificar o número de cigarros fumados/dia. (22) Além disso, a análise de perdas de seguimento foi realizada para avaliar as diferenças sistemáticas entre os participantes com dados completos e as perdas de seguimento.
Os dados são apresentados em forma de média ± dp para as variáveis cuja distribuição foi normal ou de mediana (IIQ) para aquelas cuja distribuição não foi normal. O teste t não pareado ou o teste U de Mann-Whitney foram usados para comparar as variáveis contínuas. As variáveis categóricas foram comparadas por meio do teste exato de Fisher ou do teste do qui-quadrado. O teste t pareado ou o teste de Wilcoxon foram usados para comparações referentes a um mesmo paciente. O coeficiente de correlação de Spearman foi usado para avaliar as associações entre as variáveis. As diferenças foram consideradas significativas se p < 0,05. Os dados foram analisados com o programa R, versão 3.6.2 (The R Foundation for Statistical Computing, Viena, Áustria). Gráficos e tabelas foram criados com o programa GraphPad Prism, versão 9.2 (GraphPad Software Inc., San Diego, CA, EUA).
RESULTADOS Duzentos voluntários foram avaliados para inclusão no estudo. Destes, 145 foram considerados elegíveis e foram aleatoriamente divididos em MT (n = 73) ou TCC (n = 72). Após a randomização, 28 participantes foram excluídos porque se retiraram do estudo antes da distribuição dos grupos; 2 participantes foram excluídos por causa de transtornos psiquiátricos graves não relatados durante a triagem (esquizofrenia e transtorno de pânico), e 2 foram excluídos porque estavam abstinentes no início do ensaio. No total, 113 participantes foram incluídos na análise por intenção de tratamento: 54 do grupo MT e 59 do grupo TCC (Figura 1).
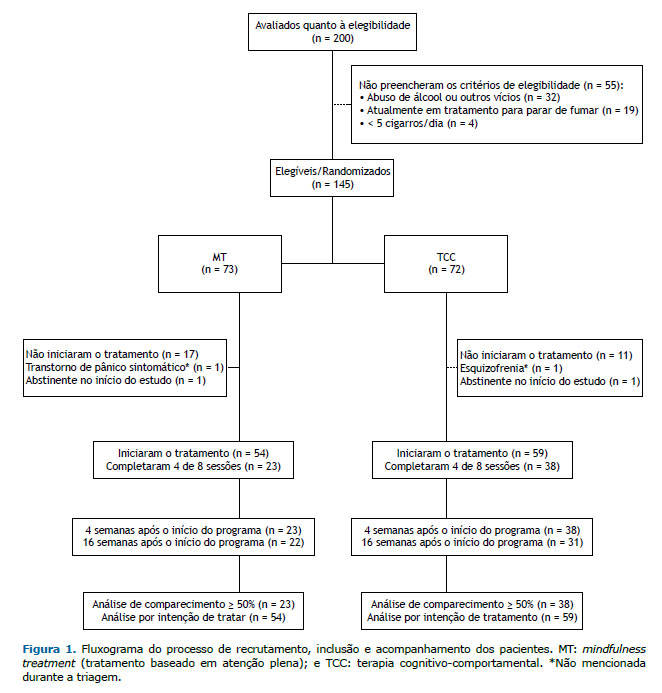
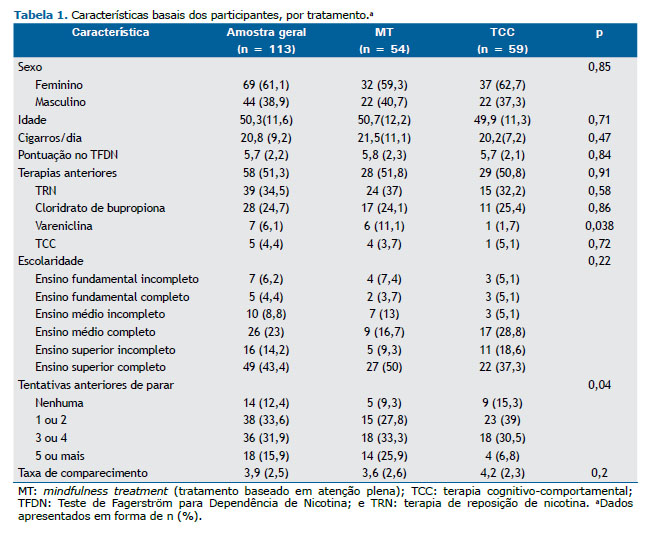
Os grupos foram semelhantes em sexo, idade, escolaridade, número de cigarros fumados/dia e pontuação obtida no TFDN. No grupo MT, o número de tentativas anteriores de cessação foi maior: 59% (vs. 32% no grupo TCC) já haviam tentado deixar de fumar três ou mais vezes. A maioria dos participantes já havia feito tratamento para parar de fumar, e o uso prévio de vareniclina foi mais comum no grupo MT (Tabela 1).
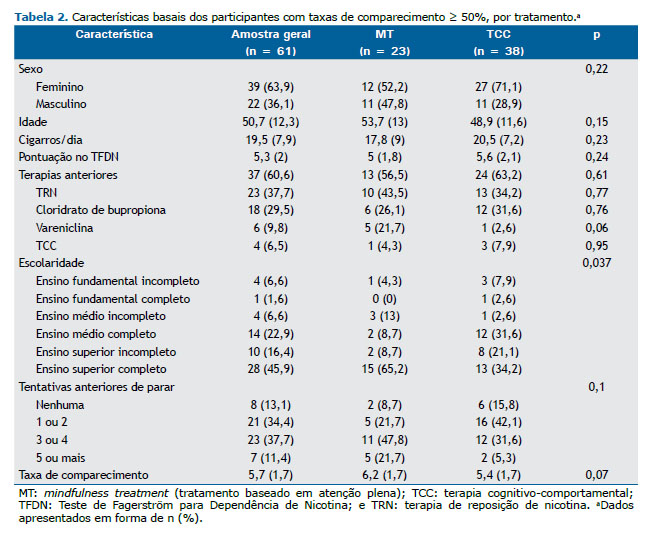
Características basais como sexo, idade, número de cigarros fumados/dia e pontuação no TFDN permaneceram semelhantes quando avaliamos o subgrupo de participantes com taxas de comparecimento ≥ 50%. A maioria (64%) dos participantes era do sexo feminino, com média de idade de 51 ± 12 anos, fumava > 20 cigarros/dia e apresentava grau moderado de dependência de nicotina (pontuação no TFDN = 5,3). Houve diferença entre os grupos quanto à escolaridade (p = 0,037): a proporção de participantes com ensino superior completo foi maior no grupo MT que no grupo TCC (65% vs. 34%; Tabela 2). Diferentemente do que foi observado na análise por intenção de tratamento, não houve diferenças entre os grupos quanto às tentativas anteriores de parar de fumar e ao uso prévio de farmacoterapia. No grupo MT, os participantes com taxas de comparecimento < 50% fumavam mais cigarros/dia antes do estudo (17,8 ± 9 vs. 24,2 ± 11,8, p = 0,03) e apresentaram pontuação maior no TFDN (6,4 ± 2,4 vs. 5 ± 1,8, p > 0,001).
Não houve diferenças significativas entre as perdas de seguimento e os participantes com dados completos quanto às características basais. Não houve diferença entre os grupos quanto ao desfecho primário (cessação do tabagismo em 16 semanas após o início do programa) e às taxas de cessação imediata (Figura 2).
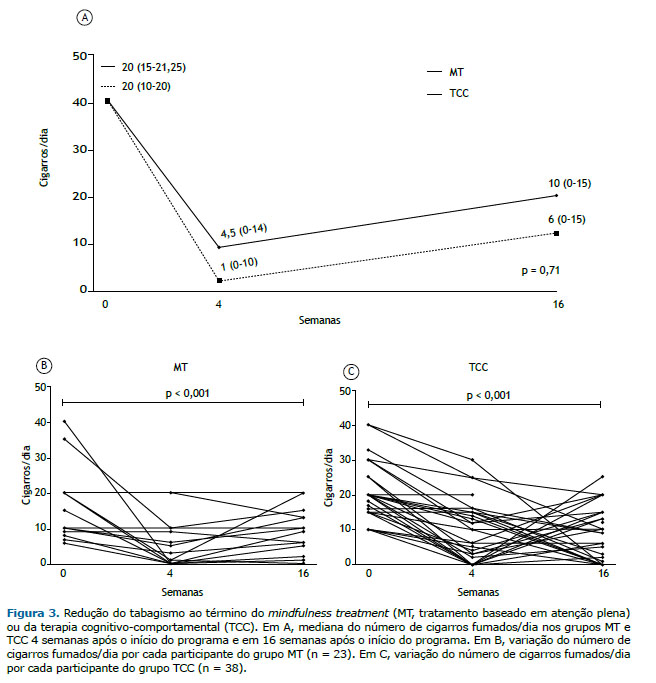
Ambos os tratamentos foram eficazes (p < 0,001) na redução do número absoluto de cigarros fumados/dia no fim do tratamento (redução de 19 cigarros no grupo MT e de 15,5 cigarros no grupo TCC) e em 16 semanas após o início do programa (redução de 14 cigarros no grupo MT e de 10 cigarros no grupo TCC). Não houve diferença entre os grupos quanto a esse desfecho (Figura 3). A redução relativa do número de cigarros fumados/dia foi = 93,3% (0-100%) no grupo MT vs. 70% (33,3-100%) no grupo TCC no fim do tratamento (p = 0,92), e 57,1% (0-100%) no grupo MT vs. 70% (25-100%) no grupo TCC em 16 semanas após o início do programa (p = 0,49).
Na análise por intenção de tratamento, não houve diferenças entre os grupos quanto à abstinência em 16 semanas após o início do programa (12,9% no grupo MT vs. 20,3% no grupo TCC, p = 0,29) e no fim do tratamento (20,4% no grupo MT vs. 23,7% no grupo TCC, p = 0,66). No entanto, quando analisamos os participantes com taxas de comparecimento ≥ 50%, ambas as terapias foram mais eficazes.
Quando avaliamos os participantes que completaram 75% dos programas (13 participantes no grupo MT vs. 14 participantes no grupo TCC), as taxas de cessação foram maiores em ambos os grupos. A taxa de cessação foi = 69% no grupo MT vs. 43% no grupo TCC (p = 0,32) no fim do tratamento, e 38% no grupo MT vs. 43% no grupo TCC (p = 0,85) em 16 semanas após o início do programa.
Houve forte correlação entre o comparecimento às sessões e a redução da proporção de cigarros fumados/dia na última sessão do programa e em 16 semanas após o início do programa (Figura 4).
DISCUSSÃO Pelo que sabemos, este é o primeiro ensaio clínico controlado randomizado a avaliar a eficácia de um MT em comparação com a TCC no Brasil. Este é também o primeiro estudo a adaptar um protocolo de MT para a cessação do tabagismo em nosso país. Os principais achados do estudo são os seguintes: 1. ausência de diferenças significativas entre os grupos quanto às taxas de abstinência ao término dos programas e em 16 semanas após o início dos programas; 2. ausência de diferenças significativas entre os grupos quanto à redução de cigarros fumados/dia ao término dos programas e em 16 semanas após o início do programa; 3. eficácia do tratamento comparável àquela relatada na literatura para ambos os tratamentos e os desfechos avaliados e 4. forte correlação entre as taxas de comparecimento às sessões e a redução de cigarros fumados/dia.
Taxas de abstinência em longo prazo de aproximadamente 16% foram alcançadas com o uso de TCC isoladamente (isto é, sem associação medicamentosa).(4) No Brasil, faltam estudos com metodologia adequada que avaliem a TCC como tratamento independente para a cessação do tabagismo. Os estudos disponíveis ou são de pequeno porte, ou não são randomizados, ou são retrospectivos ou carecem de verificação objetiva da abstinência. Não obstante, optamos por usar a TCC como intervenção de controle por vários motivos: 1) É o tratamento-padrão em nosso país; 2) Demonstrou eficácia em estudos epidemiológicos(23); 3) Possui materiais padronizados; 4) Os pneumologistas no Brasil são treinados para administrar a TCC e 5) Os componentes da TCC são bastante semelhantes aos do MT, embora os métodos propostos para alcançar a cessação sejam diferentes.
Em um estudo epidemiológico sobre programas de cessação do tabagismo baseados em TCC realizado em 2013 no estado do Paraná,(23) 54% dos participantes do estudo compareceram à quarta (e última) sessão de tratamento; destes, 41% afirmaram que estavam abstinentes (embora isso não tenha sido determinado objetivamente). Além disso, 61% também estavam recebendo tratamento farmacológico para parar de fumar.(23) No presente estudo, o grupo MT alcançou uma taxa de abstinência de 20,4% ao término do programa na análise por intenção de tratamento e uma taxa de abstinência de 47,8% quando foram avaliados apenas os participantes com taxas de comparecimento ≥ 50%. Esses resultados são comparáveis aos da literatura e são semelhantes aos do grupo controle em nosso estudo.
No presente estudo, as taxas de cessação imediata foram = 47,8% no grupo MT e 36,8% no grupo TCC (p = 0,25), ao passo que, no estudo no qual nosso protocolo de MT foi baseado, as taxas de cessação imediata foram = 36% no grupo MT e 15% no grupo TCC (p = 0,063).(11) No tocante à abstinência em longo prazo, as taxas de cessação foram = 30,4% no grupo MT e 31,6% no grupo TCC (p = 0,68) no presente estudo, ao passo que, no estudo supracitado, elas foram = 31% no grupo MT e 6% no grupo TCC (p = 0,012).(11) Portanto, os resultados foram comparáveis nos dois grupos MT, porém o grupo TCC do presente estudo teve um desempenho melhor do que o do estudo supracitado.(11) Em um ensaio clínico maior, no qual TCC ou MT foram usados em associação a adesivos de nicotina, as taxas de cessação imediata foram = 42,1% no grupo MT e 39,1% no grupo TCC, e as taxas de cessação em longo prazo foram = 19,4% no grupo MT e 23,8% no grupo TCC.(12) Como em nosso estudo, não foram encontradas diferenças significativas entre as duas intervenções naquele estudo.(12) No entanto, o MT mostrou-se superior à TCC no que tange à recuperação após uma recaída,(12) um desfecho que não foi avaliado no presente estudo.
A adesão aos programas teve impacto direto nos resultados. Isso foi corroborado por uma correlação positiva entre as taxas de comparecimento e a redução do número de cigarros fumados/dia, bem como pelas taxas de cessação mais elevadas nos participantes com taxas de comparecimento ≥ 50% e 75%.
O principal ponto forte de nosso estudo é o fato de que fomos capazes de realizar um ensaio clínico controlado randomizado com uma amostra diversificada de fumantes, incluindo homens e mulheres de diferentes faixas etárias e graus de escolaridade. Outro ponto forte é o fato de que os grupos foram equiparados no que tange à duração das sessões e atividades domiciliares propostas. Além disso, a avaliação da abstinência por meio de um questionário padronizado aplicado por um entrevistador que desconhecia a intervenção e a realização de medições do monóxido de carbono no ar expirado tornou os desfechos mais confiáveis. Acreditamos também que a análise estatística baseada nas taxas de comparecimento permitiu uma compreensão melhor dos resultados.
Nosso estudo tem limitações que precisam ser mencionadas. O alto número de desistências reduziu o poder do estudo e não permitiu uma análise de subgrupos minuciosa. A saída de participantes do estudo antes da alocação das intervenções foi maior no grupo MT, o que significa que o número de participantes no grupo MT foi menor desde o início. Essa diferença acentuou-se quando foram avaliados os subgrupos com frequência mínima de 50%. Isso pode ser parcialmente explicado pelo fato de que o número de indivíduos com múltiplas tentativas de cessação, uso prévio de medicamentos antitabagismo ou ambos foi maior no grupo MT, diferença essa que desapareceu no subgrupo com taxas de comparecimento ≥ 50%. Outra hipótese é a de que o MT pode ser mais fácil de aplicar em indivíduos com maior escolaridade, pois o conhecimento a respeito da meditação é mais difundido nessa população.(24) Em nosso estudo, os grupos exibiam escolaridade semelhante na análise por intenção de tratamento. No entanto, o grupo MT apresentou maior prevalência de indivíduos com maior escolaridade quando analisamos o subgrupo de pacientes com frequência ≥ 50%. Além disso, houve mais desistências entre os indivíduos do grupo MT que fumavam mais e apresentaram pontuação maior no TFDN, situações nas quais geralmente se recomenda associação de tratamento medicamentoso.(25)
As taxas de dados faltantes em virtude da perda de seguimento em longo prazo foram maiores no grupo TCC que no grupo MT (18% vs. 4%). Como a recidiva é comum nesse cenário, acreditamos que a análise last observation carried forward possa ter subestimado o número de cigarros consumidos por alguns desses participantes, favorecendo, portanto, o grupo TCC.
Outra possível limitação é que os instrutores de MT tinham pouca experiência com a terapia aplicada. Embora contássemos com um instrutor habilitado em atenção plena e um pneumologista com experiência em cessação do tabagismo, esta foi nossa primeira experiência com a implantação de um protocolo baseado em atenção plena para a cessação do tabagismo. É possível que profissionais com mais experiência e sessões de manutenção tivessem alcançado resultados ainda melhores.
Por fim, os resultados do presente estudo indicam que o MT é tão eficaz quanto a TCC para a cessação ou redução do tabagismo. Se considerarmos que a dependência de nicotina é uma doença muito difícil de tratar que afeta milhões de pessoas de diferentes contextos socioeconômicos, educacionais e culturais, podemos entender o quão importante é ter diferentes opções de tratamento. Como o MT usa uma estratégia significativamente diferente da TCC, ele pode se tornar uma alternativa interessante, especialmente em indivíduos que ainda não conseguiram deixar de fumar com o tratamento-padrão.
Estudos multicêntricos com amostras maiores são necessários para compreender completamente o potencial do MT para a cessação do tabagismo. Esses estudos também serão importantes para a identificação dos subgrupos de pacientes que provavelmente se beneficiarão com essa intervenção e para a investigação do uso de MT em conjunto com farmacoterapia.
AGRADECIMENTOS Em primeiro lugar, gostaríamos de agradecer aos participantes deste ensaio. Gostaríamos também de agradecer a Giovanna Lemes Leão, Isabela De Bortoli, Rafaela Portiolli Tümmler e Rebecca Saray Marchesini Stival seu papel na avaliação dos desfechos deste estudo. Finalmente, gostaríamos de expressar nossa gratidão a Judson Brewer por ter gentilmente compartilhado conosco seu Mindfulness Training Manual for Smoking Cessation (Manual de Treinamento de Atenção Plena para Cessação do Tabagismo).
CONTRIBUIÇÕES DOS AUTORES MSA, LGS e GMAP: concepção e planejamento do estudo; interpretação das evidências; redação e revisão das versões preliminares e da versão final; aprovação da versão final. NFP: concepção e planejamento do estudo; redação e revisão das versões preliminares e da versão final; aprovação da versão final. FMC, LM, DN e MC: planejamento do estudo; revisão da versão final; aprovação da versão final. MH: interpretação das evidências; revisão da versão final; aprovação da versão final.
CONFLITO DE INTERESSES Nenhum conflito declarado.
REFERÊNCIAS 1. World Health Organization [homepage on the Internet]. Geneva: World Health Organization; c2021 [updated 2021 Jul 26; cited 2021 Jun 26]. Tobacco. https://www.who.int/news-room/fact-sheets/detail/tobacco
2. Brasil: Ministério da Saúde, Secretaria de Vigilância em Saúde. VIGITEL BRASIL 2019, Vigilância de fatores de risco e proteção para doenças crônicas por inquérito telefônico. [monograph on the Internet]. Brasília: o Ministério; 2020 [cited 2021 Jun 26]. Available from: http://www.crn1.org.br/wp-content/uploads/2020/04/vigitel-brasil-2019-vigilancia-fatores-risco.pdf?x53725
3. Pinto, M, Bardach, A, Palacios, A, Biz, A, Alcaraz, A, Rodriguez, B, et al. Burden of smoking in Brazil and potential benefit of increasing taxes on cigarettes for the economy and for reducing morbidity and mortality. Cad Saude Publica. 2019;35(8):e00129118. https://doi.org/10.1590/0102-311x00129118
4. Fiore MC. US public health service clinical practice guideline: treating tobacco use and dependence. Respir Care. 2000;45(10):1200-1262. PMID: 11054899
5. Hughes JR, Keely J, Naud S. Shape of the relapse curve and long-term abstinence among untreated smokers. Addiction. 2004;99(1):29-38. https://doi.org/10.1111/j.1360-0443.2004.00540.x
6. Carlson LE, Taenzer P, Koopmans J, Bultz BD. Eight-year follow-up of a community-based large group behavioral smoking cessation intervention. Addict Behav. 2000;25(5):725-741. https://doi.org/10.1016/S0306-4603(00)00081-2
7. Terzi AM, Matos DP, Rodrigues ML, Demarzo M. Mindfulness in Education and Paulo Freire: a reflective approach. Interface (Botucatu). 2020; 24:e200015. https://doi.org/10.1590/interface.200015
8. Kabat-Zinn J, Lipworth L, Burney R. The clinical use of mindfulness meditation for the self-regulation of chronic pain. J Behav Med. 1985;8(2):163-190. https://doi.org/10.1007/BF00845519
9. Hofmann SG, Sawyer AT, Witt AA, Oh D. The effect of mindfulness-based therapy on anxiety and depression: A meta-analytic review. J Consult Clin Psychol. 2010;78(2):169-183. https://doi.org/10.1037/a0018555
10. Warren JM, Smith N, Ashwell M. A structured literature review on the role of mindfulness, mindful eating and intuitive eating in changing eating behaviours: effectiveness and associated potential mechanisms. Nutr Res Rev. 2017;30(2):272-283. https://doi.org/10.1017/S0954422417000154
11. Brewer JA, Mallik S, Babuscio TA, Nich C, Johnson HE, Deleone CM, et al. Mindfulness training for smoking cessation: results from a randomized controlled trial. Drug Alcohol Depend. 2011;119(1-2):72-80. https://doi.org/10.1016/j.drugalcdep.2011.05.027
12. Vidrine JI, Spears CA, Heppner WL, Reitzel LR, Marcus MT, Cinciripini PM, et al. Efficacy of mindfulness-based addiction treatment (MBAT) for smoking cessation and lapse recovery: A randomized clinical trial. J Consult Clin Psychol. 2016;84(9):824-838. https://doi.org/10.1037/ccp0000117
13. Oikonomou MT, Arvanitis M, Sokolove RL. Mindfulness training for smoking cessation: A meta-analysis of randomized-controlled trials. J Health Psychol. 2017;22(14):1841-1850. https://doi.org/10.1177/1359105316637667
14. Cropley M, Ussher M, Charitou E. Acute effects of a guided relaxation routine (body scan) on tobacco withdrawal symptoms and cravings in abstinent smokers. Addiction. 2007;102(6):989-993. https://doi.org/10.1111/j.1360-0443.2007.01832.x
15. Leventhal AM, Zvolensky MJ. Anxiety, depression, and cigarette smoking: a transdiagnostic vulnerability framework to understanding emotion-smoking comorbidity. Psychol Bull. 2015;141(1):176-212. https://doi.org/10.1037/bul0000003
16. Sancho M, De Gracia M, Rodríguez RC, Mallorquí-Bagué N, Sánchez-González J, Trujols J, et al. Mindfulness-Based Interventions for the Treatment of Substance and Behavioral Addictions: A Systematic Review. Front Psychiatry. 2018;9:95. https://doi.org/10.3389/fpsyt.2018.00095
17. American Psychiatric Association. DSM-IV: Manual de Diagnóstico e Estatística das Perturbações Mentais. 4th ed. Lisboa: Climepsi Editores; 2002.
18. Brasil. Ministério da Saúde. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Deixando de Fumar sem Mistérios - Manual do Coordenador, 2nd ed. Rio de Janeiro: INCA; 2019. Available from: https://www.inca.gov.br/publicacoes/manuais/deixando-de-fumar-sem-misterio-manual-do-coordenador
19. Nunes DP. Desenvolvimento de uma intervenção para cessação do uso de derivados do tabaco durante internamento por acidente vascular cerebral agudo. [dissertation]. Curitiba: Universidade Federal do Paraná; 2020.
20. American Lung Association. Freedom From Smoking [homepage on the Internet]. Chicago, IL: American Lung Association; c2021 [cited 2021 Jun 26]. Resources - Information and Tools for Your Quit Journey. Available from: https://freedomfromsmoking.org/resources/
21. Gariti P, Alterman AI, Ehrman R, Mulvaney FD, O’Brien CP. Detecting smoking following smoking cessation treatment. Drug Alcohol Depend. 2002;65(2):191-196. https://doi.org/10.1016/S0376-8716(01)00162-4
22. Hedeker D, Mermelstein RJ, Demirtas H. Analysis of binary outcomes with missing data: missing = smoking, last observation carried forward, and a little multiple imputation. Addiction. 2007;102(10):1564-1573. https://doi.org/10.1111/j.1360-0443.2007.01946.x
23. Reichert J. Seção 1, Contexto Atual. In: Reichert J. 35 anos de história da luta contra o tabagismo no Paraná. Curitiba: Secretaria de Estado da Saúde do Paraná; 2015. p.118.
24. Macinko J, Upchurch DM. Factors Associated with the Use of Meditation, U.S. Adults 2017. J Altern Complement Med. 2019;25(9):920-927. https://doi.org/10.1089/acm.2019.0206
25. Ministério da Saúde, Instituto Nacional de Câncer, Coordenação de Prevenção e Vigilância. Abordagem e Tratamento do Fumante - Consenso 2001. Capítulo 1, Bases teóricas para as recomendações dos métodos de cessação de fumar. Rio de Janeiro: INCA; 2001. p. 16.







