ABSTRACT
Objective: Many patients with proportional reductions in FVC and FEV1 on spirometry show no reduction in TLC. The aim of this study was to evaluate the role that measuring lung volumes and airway resistance plays in the correct classification of patients with a possible restrictive pattern on spirometry. Methods: This was a prospective study involving adults with reduced FVC and FEV1, as well as an FEV1/FV(C) ratio within the predicted range. Restrictive lung disease (RLD) was characterized by TLC below the 5th percentile, as determined by plethysmography. Obstructive lung disease (OLD) was characterized by high specific airway resistance, significant changes in post-bronchodilator FEV1, or an FEF25-75% < 50% of predicted, together with a high RV/TLC ratio. Nonspecific lung disease (NLD) was characterized by TLC within the predicted range and no obstruction. Combined lung disease (CLD) was characterized by reduced TLC and findings indicative of airflow obstruction. Clinical diagnoses were based on clinical suspicion, a respiratory questionnaire, and the review of tests of interest. Results: We included 300 patients in the study, of whom 108 (36%) were diagnosed with RLD. In addition, 120 (40%) and 72 (24%) were diagnosed with OLD/CLD and NLD, respectively. Among the latter, 24 (33%) were clinically diagnosed with OLD. In this sample, 151 patients (50.3%) were obese, and obesity was associated with all patterns of lung disease. Conclusions: Measuring lung volumes and airway resistance is often necessary in order to provide an appropriate characterization of the pattern of lung disease in patients presenting with a spirometry pattern suggestive of restriction. Airflow obstruction is common in such cases.
Keywords:
Spirometry; Airway resistance; Lung volume measurements.
RESUMO
Objetivo: Muitos pacientes com redução proporcional de CVF e VEF1 na espirometria não têm CPT reduzida. O objetivo deste estudo foi avaliar o papel da medida dos volumes pulmonares e da resistência das vias aéreas para a classificação correta de pacientes com possível restrição à espirometria. Métodos: Estudo prospectivo de adultos com CVF e VEF1 reduzidos e relação VEF1/CV(F) na faixa prevista. Distúrbio ventilatório restritivo (DVR) foi definido por CPT < 5º percentil por pletismografia. Distúrbio ventilatório obstrutivo (DVO) foi caracterizado por resistência específica de vias aéreas elevada, resposta significativa do VEF1 pós-broncodilatador e/ou um FEF25-75% < 50% do previsto associado a uma relação VR/CPT elevada. Distúrbio ventilatório inespecífico (DVI) foi caracterizado por CPT na faixa prevista e ausência de obstrução. Distúrbio ventilatório combinado (DVC) foi caracterizado por CPT reduzida e achados indicativos de obstrução ao fluxo aéreo. Os diagnósticos clínicos foram baseados em suspeita clínica, um questionário respiratório e revisão de exames de interesse. Resultados: Foram incluídos 300 pacientes no estudo, dos quais 108 (36%) tiveram diagnóstico de DVR, enquanto 120 (40%) foram diagnosticados com DVO ou DVC e 72 (24%) com DVI. Destes últimos, 24 (33%) tinham diagnóstico clínico de DVO. Nesta amostra, 151 pacientes (50,3%) eram obesos, e isso se associou com todos os padrões de distúrbios funcionais. Conclusões: Medidas dos volumes pulmonares e da resistência das vias aéreas são frequentemente necessárias para a caracterização adequada do tipo de distúrbio funcional em casos com possível restrição à espirometria. A obstrução ao fluxo aéreo é comum nesses casos.
Palavras-chave:
Espirometria; Resistência das vias respiratórias, Medidas de volume pulmonar.
INTRODUCTIONThe American Thoracic Society (ATS)/European Respiratory Society (ERS) task force proposed definitions for the various patterns of lung disease.(1) Restrictive lung disease (RLD) was defined as a reduction in TLC below the 5th percentile of the predicted value and a normal FEV1/VC ratio. Obstructive lung disease (OLD) was defined as an FEV1/VC ratio below the 5th percentile of the predicted value. Mixed or combined lung disease (CLD) was character-ized by FEV1/VC and TLC below the 5th percentile of the predicted values.
A combination of reduced VC and preserved FEV1/(F)VC is used in order to infer the presence of RLD; however, in approximately 40% of such cases, TLC is not reduced.(2,3) According to the ATS/ERS task force, OLD is characterized by a combination of reduced (F)VC, FEV1/(F)VC above the lower limit of normal, and TLC within the predicted range.(1) This functional abnormality was later designated nonspecific lung disease (NLD).(4) In a sample of 100 patients pre-senting with reduced (F)VC, FEV1/(F)VC above the lower limit of normal, and TLC within the predicted range, 68 had evidence of airway disease, whereas the remaining 32 had signs of restriction.(4)
The proportional reduction in FVC and FEV1 in patients with OLD can be explained by airway closure with air trap-ping.(5) Obesity reduces (F)VC more than it does FEV1(6) and can therefore result in a preserved FEV1/(F)VC ratio in the presence of OLD. As occurs with diseases affecting respiratory mechanics or respiratory muscle strength, obesity can, in and of itself, result in NLD.(4) Although COPD and asthma account for most OLDs, a wide range of other diseases, including bronchiolar diseases and some interstitial lung diseases, are associated with airflow obstruction and can result in proportional reductions in FVC and FEV1.(7) In addition, smoking (either current or past) is associated with various lung diseases and can contribute to an obstructive component.
Spirometry is considered the method of choice for detecting airflow limitation caused by OLD. However, airflow lim-itation is multifactorial. One such factor is high airway resistance (Raw).(8) In many patients with spirometry results suggestive of RLD, Raw measurements can reveal airflow obstruction. It is commonly believed that Raw is a parame-ter that is not sufficiently sensitive in cases of peripheral airway disease; however, a classic study showed a close correlation between airway conductance (Gaw) and bronchiolar diameter.(9) It is possible that Gaw alone is abnormal in patients with bronchiolitis. (10) In 2012, reference values for specific Raw were derived from a large sample of healthy adults.(11)
The objective of the present study was to evaluate the role that measuring lung volumes and Raw plays in the final functional classification of patients with spirometry results suggestive of RLD.
METHODSData collection was performed in the pulmonary function laboratories of Centro Diagnóstico Brasil (n = 217) and the São Paulo Hospital for State Civil Servants (n = 83) in the period between December of 2011 and December of 2013. Pulmonologists certified in pulmonary function testing by the Brazilian Thoracic Association (BTA) and the lead author of the present study prospectively selected all spirometry results suggestive of RLD. Clinical diagnosis was established by the pulmonologist requesting the test, by administering a standardized respiratory questionnaire adapted from a previously published questionnaire (Appendix 1: http://www.jornaldepneumologia.com.br/detalhe_anexo.asp?id=46)(12) and by reviewing ancillary test results or analyzing the results of additional tests, including chest X-rays, chest CT scans, and echocardiograms, requested on the basis of clinical suspicion. All pulmonary function tests were performed in accordance with the BTA guidelines.(13) All patients gave written informed consent.
Inclusion criteriaThe inclusion criteria were as follows: 1) being an adult whose age and height were within the reference range(14); 2) having FVC below the lower limit of normal, i.e., below the 5th percentile of the reference population(14); 3) having FEV1/FVC and FEV1/VC equal to or above the lower limit of normal, i.e., above the 5th percentile of the reference population(14); 4) having a definitive clinical diagnosis (for asthma, physician-diagnosed asthma and a patient report of two or more episodes of wheezing, which were alleviated by bronchodilator use; for COPD, physician-diagnosed COPD, chronic cough/dyspnea-a Medical Research Council scale score ≥ 2-and past or current smoking; patients diagnosed with obesity were in most cases referred for preoperative evaluation for bariatric surgery, including those with a complaint of dyspnea without meeting criteria for diseases such as asthma); and 5) having performed pulmo-nary function tests in accordance with the BTA/ATS/ERS acceptability and reproducibility criteria.(13,15-17)
Patients whose tests were not in accordance with the aforementioned criteria were excluded, as were those without a definitive diagnosis by the end of the analysis period.
All pulmonary function tests were performed with a Sensor Medics 6200 Bodybox system and a Collins system (Fer-raris Respiratory, Louisville, CO, USA). Lung volumes were determined by whole-body plethysmography. For lung volumes, the predicted values were those proposed by Crapo et al.(18) Reduced TLC was characterized by values below the 5th percentile. RV and the RV/TLC ratio were considered high when they were above the 95th percentile of the reference values.(18) Spirometry was repeated after administration of a bronchodilator (400 µg of albuterol aero-sol). A significant bronchodilator response was characterized by FEV1 ≥ 0.20 L and 7% of predicted, in accordance with Soares et al.(19)
Raw was measured by mean linear intercept values, as recommended by Matthys et al., after analysis of at least five pressure-flow loops.(20) Only satisfactory, reproducible loops were accepted. The predicted values used for calcu-lation were those proposed by Piatti et al.(11) Values above 8.0 cmH2O/s in females and 8.6 cmH2O/s in males were considered high (mean ± 1.64 SD).
Satisfactory single-breath DLCO measurements were obtained in 260 patients. The reference values were based on those proposed by Miller et al.(21)
After data collection, the patterns of lung disease were divided into four groups:
RLD-characterized by TLC below the lower limit of normal and no obstruction(1)
OLD-characterized by one or more of the following: high specific Raw corrected for lung volume (Raw × Lv); a significant change in FEV1 after bronchodilator administration (ΔFEV1 > 0.20 L and 7% of predicted); and FEF25-75% < 50% of predicted with a high RV/TLC ratio (see the Results section)
CLD-characterized by reduced TLC and findings indicative of airflow obstruction, including high Raw × Lv; FEF25-75% < 50% with a high RV/TLC ratio; and a significant bronchodilator response
NLD-characterized by TLC within the predicted range and no functional findings indicative of obstruction
All values were expressed as mean ± standard deviation. The groups were compared by means of the Student's t-test and ANOVA (for continuous independent variables), and the chi-square test (for nominal variables). Correlations between Raw × Lv and functional parameters were determined by Spearman's test. The distribution of Raw × Lv was lognormal, and Raw × Lv values were transformed for comparison. ROC curve analysis was used in order to corre-late functional parameters and the RV/TLC ratio with specific Raw. Statistical analysis was performed with the IBM SPSS Statistics software package, version 20 (IBM Corp., Armonk, NY, USA). The level of significance was set at α = 0.05.
RESULTSA total of 300 patients were included in the present study. Table 1 shows the general characteristics of the 300 pa-tients included in the study, and Table 2 shows pulmonary function test results expressed as mean ± SD.

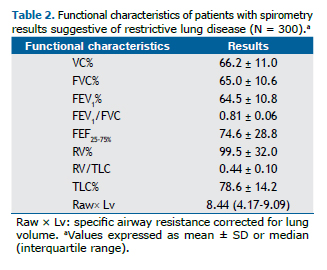
Clinical diagnoses were divided into four groups: obstructive diseases, interstitial diseases, obesity, and other dis-eases (Figure 1).
Of the 300 patients included in the present study, 151 (50.3%) were obese, but only 52 (17.3%) had a final diagno-sis of obesity without other conditions. In addition, 172 (57.3%) had TLC below the lower limit of normal (RLD), and 128 (42.7%) had TLC within the predicted range (n = 127) or high TLC (n = 1).
RV and the RV/TLC ratio were above the upper limit of normal in 46 (15.3%) and 126 (42.0%), respectively. High Raw × Lv was observed in 97 patients (32.3%). Raw × Lv (and Gaw/Lv) correlated more strongly with FEF25-75% (rs = 0.55) than with FEV1/FVC (rs = 0.50) or percent predicted FEV1 (rs = 0.27; p < 0.01 for all). Raw × Lv also correlated significantly with the RV/TLC ratio (rs = 0.46; p < 0.001). ROC curve analysis showed that the area under the ROC curve was higher for FEF25-75% than for FEV1/FVC or percent predicted FEV1 (i.e., 0.75; p < 0.001) for differentiating between patients with and without high Raw × Lv. An FEF25-75% of less than 50% had a sensitivity of 40% and a speci-ficity of 89% for detecting high specific Raw. With regard to lung volume measurements, the RV/TLC ratio had the highest area under the curve for characterizing airflow obstruction (0.75; p < 0.01). Given that a high RV/TLC ratio and FEF25-75% < 50% can each be found in patients with RLD or NLD, they were combined in order to characterize airflow obstruction. A combination of high RV/TLC and FEF25-75% < 50% was found in 46 patients. In 14 of those, the aforementioned parameters constituted the only evidence of obstruction.
A significant bronchodilator response was observed in 23 patients (7.7%). The most common clinical diagnoses in those patients were obstructive diseases (n = 12) and obesity (n = 5). Of the 14 patients diagnosed with congestive heart failure (CHF), only 1 (7.0%) had a significant bronchodilator response.
On the basis of one or more of the aforementioned criteria, 120 patients (40.0%) had airflow obstruction. Of the 120 patients with OLD, 64 (53.3%) had TLC below the lower limit of normal and were therefore considered to have CLD. Of the 128 patients with TLC within the predicted range, 72 (56.2%) had no airflow obstruction and were therefore classified as having NLD.
All patterns of lung disease found in the present study and the respective clinical diagnoses are shown in Figure 2. In the four groups of lung diseases there were patients diagnosed with obesity and patients diagnosed with other diseases. Four of the patients who were diagnosed with asthma had RLD, and 17 of the patients in the CLD group had a diagnosis of OLD, asthma being the most common obstructive disease (n = 10).
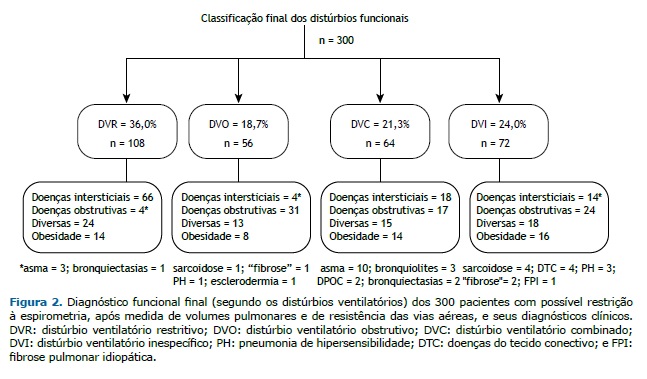
Of the 72 patients who were diagnosed with NLD, 24 (33.3%) had a clinical diagnosis of obstructive disease: asth-ma, in 11; COPD, in 8; bronchiectasis, in 3; and bronchiolitis, in 3. Therefore, of the 300 patients included in the present study, 144 (i.e., the aforementioned 24 plus the 120 who were diagnosed with OLD or CLD, accounting for 48.0% of the sample) had obstructive disease.
Several variables, including age, gender, and functional results, were compared among the four major clinical di-agnostic groups (Table 3). The patients with a final diagnosis of obesity were younger and had a body mass index above 35 kg/m2 (79%); therefore, most were referred for preoperative evaluation for bariatric surgery. Expiratory reserve volume (ERV) was lowest in the group of patients who were diagnosed with obesity, who, in comparison with the group of patients diagnosed with interstitial diseases, had higher percent predicted VC, FVC, RV, and TLC; more preserved DLCO; and similar Raw × Lv.
 DISCUSSION
DISCUSSIONThe present study confirms the findings of previous studies(2-4) showing that spirometric findings of reduced FVC and FEV1 associated with a preserved FEV1/FVC ratio are of limited value in establishing a functional diagnosis. In addition, the study shows that lung volume and Raw measurements provide a more consistent functional characteri-zation.
It is widely recognized that adequate expiratory effort and time are required in order to characterize reduced FVC on spirometry. Incomplete exhalation often results in a restrictive pattern on spirometry. In the present study, all tests were carefully performed and reviewed.
The ATS/ERS task force defined RLD as a reduction in TLC below the 5th percentile of the predicted value and a normal FEV1/FVC ratio.(1) The reference values for TLC are therefore of great importance. In the present study, we used the values proposed by Crapo et al.(18) Although one study derived reference values for lung volumes in a sample of healthy adults in Brazil,(22) the number of individuals included in that study was small. We cannot exclude the possibility that patients with obstructive diseases that were classified as having RLD or CLD in the present study would have been better classified had there been a more suitable equation for calculating predicted lung volumes. However, cases of asthma with true restriction (reduced TLC) not associated with obesity have been described in the literature, including those with changes in lung function.(23) Such cases are sometimes encountered in clinical prac-tice.
RLD can be due to interstitial diseases, such as pulmonary fibrosis; non-respiratory conditions that secondarily im-pede lung expansion, such as muscle weakness, pleural diseases, obesity, and kyphoscoliosis; and conditions that directly affect lung function, such as CHF. Several studies have found the prevalence of spirometry-defined RLD to be 7-14%.(24-26) The prevalence of RLD is higher in males, heavy smokers, elderly individuals, individuals with a lower level of education, individuals with diabetes, individuals with CHF, and individuals with a very low or very high body mass index. Heavy smokers commonly have smoking-related interstitial lung disease, which can result in RLD or CLD.(27)
The obesity epidemic does not spare developing countries. For several reasons, obesity introduces confounding factors in the interpretation of lung function. Obesity affects lung volume measurements and spirometric values, particularly by reducing ERV and, consequently, functional residual capacity.(28) In the present study, ERV was signif-icantly lower in the group of patients diagnosed with obesity. Proportional reductions in FVC and FEV1 resulting in a preserved or slightly increased FEV1/FVC ratio have been reported in obese individuals. However, although statisti-cally significant, reductions in FVC and FEV1 are typically small, and FEV1, FVC, and TLC usually remain within the range of predicted values.(6,28)
In our study, obese individuals constituted half of the sample. Obesity was found in patients with RLD, OLD, CLD, and NLD. The interaction among obesity, lung function, asthma, and COPD has been the subject of several studies and excellent reviews.(6,29-31) Obesity is associated with an increased risk of asthma.(30,31) In obese individuals, dyspnea can be attributed to obesity itself or asthma, resulting in overdiagnosis and underdiagnosis.(32) Methacholine challenge testing is useful in such cases.(4)
Lung volume measurements can aid in differentiating between RLD and OLD in patients with spirometry results suggestive of restriction.(1) One study showed that the level of agreement between clinical diagnosis and diagnosis based on pulmonary function test results (including lung volume measurements) is low and therefore does not allow differentiation between RLD and OLD.(33)
In the present study, Raw measurements and the combination of FEF25-75% and a high RV/TLC ratio allowed the diag-nosis of airflow obstruction in several cases, the level of agreement between that diagnosis and clinical diagnosis being significant. Given that the total cross-sectional area of the airways decreases dramatically from the periphery to the central regions of the lung, Raw measurements are theoretically less sensitive to peripheral changes. However, measurements of specific Raw can be useful. One study showed that Gaw measurements were more sensitive than spirometry for detecting airflow obstruction in patients with bronchiolitis obliterans syndrome.(10)
In patients with COPD, in whom obstruction is peripheral and mostly mild, the FEV1/FVC ratio is low and Raw or specific Gaw is within the predicted range; however, in the pulmonary function laboratory setting, the opposite has also been observed. A study conducted nearly 30 years ago showed that a combination of clinical and whole-body plethysmography data detected 18% of airflow obstruction cases.(34) In a classic study of 26 postmortem lungs from sudden death victims, a nearly perfect hyperbolic correlation was found between mean bronchiolar diameter and Raw (r = 0.89), whereas the correlation between mean segmental bronchial diameter and Raw was not significant.(9) In the present study, high Raw × Lv was significantly associated with reduced FEF25-75% and increased RV/TLC, sug-gesting a correlation with peripheral airway obstruction. All patients had an FEV1/FVC ratio within the predicted range.
As occurred with obesity, a final diagnosis of "other diseases" was made in patients with RLD, OLD, CLD, and NLD. Diseases included CHF, pleural disease, chest wall disease (particularly kyphoscoliosis), and neuromuscular disease. Of the 102 patients with interstitial lung disease, only 66 (65.0%) had RLD alone, as confirmed by TLC measure-ments. The remaining 36 had OLD alone (n = 4), CLD (n = 18), or NLD (n = 14). Combined pulmonary fibrosis and emphysema is a relatively common condition, given that both are smoking-related diseases.(35) In patients with connective tissue disease, bronchiolitis and emphysema associated with interstitial disease and muscle weakness can result in OLD and NLD, respectively.(36,37) In patients with hypersensitivity pneumonia or sarcoidosis, airway involvement is common and can result in OLD.(38,39)
In a study by Hyatt et al.,(4) 68% of the patients with NLD had a final diagnosis of OLD, which is in accordance with the ATS/ERS guidelines stating that proportional reductions in FVC and FEV1 with TLC within the predicted range are indicative of OLD. However, many of the patients with airway disease had a reduced FEV1/slow VC ratio. In the pre-sent study, such cases were excluded, and, as a result, only one third of all NLD patients were clinically diagnosed with OLD.
Our sample selection strategy limits generalizability of results. Because of the large number of patients routinely treated at the study facilities, selected patients were not consecutive. It is possible that there was discrepancy be-tween functional and clinical diagnoses, given that not all tests for other causes of RLD were performed. However, we believe that the objective of the present study was achieved.
In conclusion, lung volume and Raw measurements are often necessary in order to provide an appropriate charac-terization of the pattern of lung disease in patients with spirometry results suggestive of restriction. Diseases accom-panied by airflow obstruction can result in a restrictive pattern on spirometry.
REFERENCES1. Pellegrino R, Viegi G, Brusasco V, Crapo RO, Burgos F, Casaburi R, et al. Interpretative strategies for lung function tests. Eur Respir J. 2005;26(5):948-68. http://dx.doi.org/10.1183/09031936.05.00035205
2. Aaron SD, Dales RE, Cardinal P. How accurate is spirometry at predicting restrictive pulmonary impairment? Chest. 1999;115(3):869-73. http://dx.doi.org/10.1378/chest.115.3.869
3. Venkateshiah SB, Ioachimescu OC, McCarthy K, Stoller JK. The utility of spirometry in diagnosing pulmonary restriction. Lung. 2008;186(1):19-25. http://dx.doi.org/10.1007/s00408-007-9052-8
4. Hyatt RE, Cowl CT, Bjoraker JA, Scanlon PD. Conditions associated with an abnormal nonspecific pattern of pulmonary function tests. Chest. 2009;135(2):419-24. http://dx.doi.org/10.1378/chest.08-1235
5. Stănescu D, Veriter C. A normal FEV1/VC ratio does not exclude airway obstruction. Respiration. 2004;71(4):348-52. http://dx.doi.org/10.1159/000079638
6. Salome MC, King GG, Berend N. Effects of obesity on lung function. In: Dixon AE, Clerisme-Beaty EM, editors. Obesity and lung disease: a guide to management. New York: Springer Science; 2013. p. 1-20. http://dx.doi.org/10.1007/978-1-62703-053-3_1
7. Ryu JH, Scanlon PD. Obstructive lung diseases: COPD, asthma, and many imitators. Mayo Clin Proc. 2001;76(11):1144-53. http://dx.doi.org/10.4065/76.11.1144
8. Kaminsky DA. What does airway resistance tell us about lung function? Respir Care. 2012;57(1):85-96. http://dx.doi.org/10.4187/respcare.01411
9. Niewoehner DE, Kleinerman J. Morphologic basis of pulmonary resistance in the human lung and effects of aging. J Appl Physiol. 1974;36(4):412-8.
10. Bassiri AG, Girgis RE, Doyle RL, Theodore J. Detection of small airway dysfunction using specific airway conductance. Chest. 1997;111(6):1533-5. http://dx.doi.org/10.1378/chest.111.6.1533
11. Piatti G, Fasano V, Cantarella G, Tarantola C. Body plethysmographic study of specific airway resistance in a sample of healthy adults. Respirology. 2012;17(6):976-83. http://dx.doi.org/10.1111/j.1440-1843.2012.02206.x
12. Aguiar VA, Beppu OS, Romaldini H, Ratto OR, Nakatani J. Validity of a respiratory modified questionnaire (ATS-DLS-78) as a tool of an epidemiologic study in Brazil [Article in Portuguese]. J Pneumol. 1988;14(3):111-6.
13. Sociedade Brasileira de Pneumologia e Tisiologia. Diretrizes para testes de função pulmonar. J Pneumol. 2002;28(Suppl 3):S1-S238.
14. Pereira CA, Sato T, Rodrigues SC. New reference values for forced spirometry in white adults in Brazil. J Bras Pneumol. 2007;33(4):397-406. http://dx.doi.org/10.1590/S1806-37132007000400008
15. Pereira CA. Espirometria. J Pneumol. 2002;28(Suppl 3)S1-S82.
16. Standardization of Spirometry, 1994 Update. American Thoracic Society. Am J Respir Crit Care Med. 1995;152(3):1107-36. http://dx.doi.org/10.1164/ajrccm.152.3.7663792
17. Wanger J, Clausen JL, Coates A, Pedersen OF, Brusasco V, Burgos F, et al. Standardisation of the measurement of lung volumes. Eur Respir J. 2005;26(3):511-22. http://dx.doi.org/10.1183/09031936.05.00035005
18. Crapo RO, Morris AH, Clayton PD, Nixon CR. Lung volumes in healthy nonsmoking adults. Bull Eur Physiopathol Respir. 1982;18(3):419-25.
19. Soares AL, Pereira CA, Rodrigues SC. Spirometric changes in obstructive disease: after all, how much is significant? J Bras Pneumol. 2013;39(1):56-62. http://dx.doi.org/10.1590/S1806-37132013000100008
20. Matthys H, Orth U. Comparative measurements of airway resistance. Respiration.1975;32(2):121-34. http://dx.doi.org/10.1159/000193642
21. Miller A, Thornton JC, Warshaw R, Anderson H, Teirstein AS, Selikoff IJ. Single breath diffusing capacity in a representative sample of the population of Michigan, a large industrial state. Predicted values, lower limits of normal, and frequencies of abnormality by smoking history. Am Rev Respir Dis. 1983;127(3):270-7.
22. Neder JA, Andreoni S, Castelo-Filho A, Nery LE. Reference values for lung function tests. I. Static volumes. Braz J Med Biol Res. 1999;32(6):703-17. http://dx.doi.org/10.1590/s0100-879x1999000600006
23. Miller A, Palecki A. Restrictive impairment in patients with asthma. Respir Med. 2007; 101(2):272-6. http://dx.doi.org/10.1016/j.rmed.2006.05.008
24. Mannino DM, Ford ES, Redd SC. Obstructive and restrictive lung disease and functional limitation: data from the Third National Health and Nutrition Examination. J Intern Med. 2003;254(6),540-7. http://dx.doi.org/10.1111/j.1365-2796.2003.01211.x
25. Soriano JB, Miravitlles M, García-Río F, Muñoz L. Sánchez G, Sobradillo V, et al. Spirometrically-defined restrictive ventilatory defect: population variability and individual determinants. Prim Care Respir J. 2012;21(2):187-93. http://dx.doi.org/10.4104/pcrj.2012.00027
26. Wan ES, Hokanson JE, Murphy JR, Regan EA, Make BJ, Lynch DA, et al. Clinical and radiographic predictors of GOLD-unclassified smokers in the COPDGene study. Am J Respir Crit Care Med. 2011;184(1):57-63. http://dx.doi.org/10.1164/rccm.201101-0021OC
27. Sverzellati N, Guerci L, Randi G, Calabrò E, La Vecchia C, Marchianò A, et al. Interstitial lung diseases in a lung cancer screening trial. Eur Respir J. 2011;38(2):392-400. http://dx.doi.org/10.1183/09031936.00201809
28. Jones RL, Nzekwu MM. The effects of body mass index on lung volumes. Chest. 2006;130(3):827-33. http://dx.doi.org/10.1378/chest.130.3.827
29. Nicolacakis K, Skowronski ME, Coreno AJ, West E, Nader NZ, Smith RL, et al. Observations on the physiological interactions between obesity and asthma. J Appl Physiol (1985). 2008;105(5):1533-41. http://dx.doi.org/10.1152/japplphysiol.01260.2007
30. Brashier B, Salvi S. Obesity and asthma: physiological perspective. J Allergy (Cairo). 2013;2013:198068. http://dx.doi.org/10.1155/2013/198068
31. Brazzale DJ, Pretto JJ, Schachter LM. Optimizing respiratory function assessments to elucidate the impact of obesity on respiratory health. Respirology. 2015;20(5):715-21. http://dx.doi.org/10.1111/resp.12563
32. van Huisstede A, Castro Cabezas M, van de Geijn GJ, Mannaerts GH, Njo TL, Taube C, et al. Underdiagnosis and overdiagnosis of asthma in the morbidly obese. Respir Med. 2013;107(9):1356-64. http://dx.doi.org/10.1016/j.rmed.2013.05.007
33. Hong Y, Ra SW, Shim TS, Lim CM, Koh Y, Lee SD, et al. Poor interpretation of pulmonary function tests in patients with concomitant decreases in FEV1 and FVC. Respirology. 2008;13(4):569-74. http://dx.doi.org/10.1111/j.1440-1843.2008.01274.x
34. Gilbert R, Auchincloss JH Jr. The interpretation of the spirogram. How accurate is it for 'obstruction'? Arch Intern Med. 1985;145(9):1635-9. http://dx.doi.org/10.1001/archinte.1985.00360090103016
35. Jankowich MD, Rounds SI. Combined pulmonary fibrosis and emphysema syndrome: a review. Chest. 2012;141(1):222-31. http://dx.doi.org/10.1378/chest.11-1062
36. Cottin V, Cordier JF. Combined pulmonary fibrosis and emphysema in connective tissue disease. Curr Opin Pulm Med. 2012;18(5):418-27. http://dx.doi.org/10.1097/MCP.0b013e328356803b
37. Wells AU. Pulmonary function tests in connective tissue disease. Semin Respir Crit Care Med. 2007;28(4):379-88. http://dx.doi.org/10.1055/s-2007-985610
38. Bourke SJ, Carter R, Anderson K, Boyd J, King S, Douglas B, Boyd G. Obstructive airways disease in non-smoking subjects with pigeon fanciers' lung. Clin Exp Allergy. 1989;19(6):629-32. http://dx.doi.org/10.1111/j.1365-2222.1989.tb02758.x
39. LAOHABURANAKIT P, CHAN A. OBSTRUCTIVE SARCOIDOSIS. CLIN REV ALLERGY IMMUNOL. 2003;25(2):115-29. HTTP://DX.DOI.ORG/10.1385/CRIAI:25:2:115






