ABSTRACT
Objective: To evaluate the prevalence of sarcopenia in COPD patients, as well as to determine whether sarcopenia correlates with the severity and prognosis of COPD. Methods: A cross-sectional study with COPD patients followed at the pulmonary outpatient clinic of our institution. The patients underwent dual-energy X-ray absorptiometry. The diagnosis of sarcopenia was made on the basis of the skeletal muscle index, defined as appendicular lean mass/height2 only for low-weight subjects and adjusted for fat mass in normal/overweight subjects. Disease severity (COPD stage) was evaluated with the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria. The degree of obstruction and prognosis were determined by the Body mass index, airflow Obstruction, Dyspnea, and Exercise capacity (BODE) index. Results: We recruited 91 patients (50 females), with a mean age of 67.4 ± 8.7 years and a mean BMI of 25.8 ± 6.1 kg/m2. Sarcopenia was observed in 36 (39.6%) of the patients, with no differences related to gender, age, or smoking status. Sarcopenia was not associated with the GOLD stage or with FEV1 (used as an indicator of the degree of obstruction). The BMI, percentage of body fat, and total lean mass were lower in the patients with sarcopenia than in those without (p < 0.001). Sarcopenia was more prevalent among the patients in BODE quartile 3 or 4 than among those in BODE quartile 1 or 2 (p = 0.009). The multivariate analysis showed that the BODE quartile was significantly associated with sarcopenia, regardless of age, gender, smoking status, and GOLD stage. Conclusions: In COPD patients, sarcopenia appears to be associated with unfavorable changes in body composition and with a poor prognosis.
Keywords:
Sarcopenia; Body composition; Pulmonary disease, chronic obstructive; Severity of illness index.
RESUMO
Objetivo: Avaliar a prevalência de sarcopenia em pacientes com DPOC e determinar se sarcopenia está correlacionada com a gravidade e o prognóstico de DPOC. Métodos: Estudo retrospectivo em pacientes com DPOC atendidos no ambulatório de pneumologia de nossa instituição. Os pacientes realizaram absorciometria de dupla energia por raios X. O diagnóstico de sarcopenia foi baseado no índice de massa muscular esquelética, definido como massa magra apendicular/altura2 somente para indivíduos com baixo peso, sendo ajustado pela massa gorda para aqueles com peso normal/sobrepeso. A gravidade da doença (estádio da DPOC) foi avaliada com os critérios da Global Initiative for Chronic Obstructive Lung Disease (GOLD). O grau de obstrução e o prognóstico foram determinados pelo índice Body mass index, airflow Obstruction, Dyspnea, and Exercise capacity (BODE). Resultados: Foram incluídos 91 pacientes (50 mulheres), com média de idade de 67,4 ± 8,7 anos e média de IMC de 25,8 ± 6,1 kg/m2. Sarcopenia foi diagnosticada em 36 (39,6%) dos pacientes, sem diferenças relacionadas a sexo, idade ou status tabágico. Não houve associação de sarcopenia com estádios GOLD ou VEF1 (utilizado como indicador do grau de obstrução). O IMC, a porcentagem de gordura corporal e a massa magra total foram menores nos pacientes com sarcopenia do que naqueles sem a doença (p < 0,001). A prevalência de sarcopenia foi maior nos pacientes com BODE nos quartis 3 ou 4 que naqueles com BODE nos quartis 1 ou 2 (p = 0,009). A análise multivariada mostrou que os quartis do BODE estavam significativamente associados à sarcopenia, independentemente de idade, gênero, status tabágico e estádio GOLD. Conclusões: Em pacientes com DPOC, sarcopenia parece estar associada a alterações desfavoráveis na composição corporal e pior prognóstico.
Palavras-chave:
Sarcopenia; Composição corporal; Doença pulmonar obstrutiva crônica; Índice de gravidade de doença.
INTRODUÇÃOÉ bem sabido que a DPOC é uma doença altamente prevalente, atingindo até 10% dos adultos acima de 40 anos, com altas taxas de morbida-de e mortalidade.(1) Associa-se a diversas condições extrapulmonares, tais como doenças cardiovasculares, osteoporose, caquexia e anemia. Estudos anteriores indicam uma associação entre baixo IMC e menor sobrevida em pacientes com DPOC.(2,3) Porém, dados mais recentes de-monstram que alterações específicas e desfavoráveis na composição corporal, especialmente a diminuição da massa magra, podem ser predito-res mais confiáveis de mortalidade do que o baixo IMC por si só.(4,5) Em pacientes com DPOC, tais mudanças já foram relacionadas a intolerân-cia ao exercício, comprometimento da qualidade de vida e aumento da mortalidade.(6) Poucos estudos na literatura correlacionaram a prevalên-cia de sarcopenia a índices de gravidade da DPOC. Além disso, até o momento, não há estudos correlacionando a sarcopenia ao prognóstico da DPOC ou corrigindo a sarcopenia pelo IMC para evitar erros de diagnóstico em pacientes com sobrepeso.
Em pacientes com DPOC com mais de 50 anos de idade, há uma redução de 1-2% ao ano na massa muscular. (4) Além disso, entre aqueles na faixa etária de 50 a 60 anos e aqueles com mais de 60 anos de idade, já foi demonstrado que a força muscular declina 1,5% e 3,0% ao ano, respectivamente.(4) Esse fenômeno, conhecido como sarcopenia, é um importante indicador da síndrome de fragilidade. Já foi demonstrado que a sarcopenia ocorre em aproximadamente 5-13% de todos os indivíduos com mais de 65 anos de idade(7) e em 20-40% de todos os pacientes com DPOC, podendo incluir ainda os 10-15% de pacientes com DPOC que têm peso normal.(2)
Em pacientes com DPOC, a diminuição da capacidade de exercício é o principal fator limitante das atividades de vida diária e está diretamen-te relacionada a maior risco de exacerbações.(8) Já foi sugerido que tal diminuição é o melhor preditor de mortalidade precoce na DPOC.(3) O grau de comprometimento da capacidade de exercício (intolerância ao exercício), que resulta de fatores como comprometimento da função pulmonar, limitação das trocas gasosas e disfunção muscular esquelética, está relacionado à gravidade da DPOC. Na presença de dispneia, tais alterações levam a maior comprometimento da atividade física, iniciando um círculo vicioso, também conhecido como espiral descendente.(6,8)
O objetivo do presente estudo foi avaliar a prevalência de sarcopenia em pacientes com DPOC. Também se procurou determinar se a sarco-penia está correlacionada com os índices de gravidade da DPOC e com o prognóstico da mesma.
MÉTODOSPacientes e procedimentosTrata-se de um estudo transversal com 96 pacientes consecutivos com DPOC, todos com mais de 50 anos de idade, atendidos no Ambulatório de Pneumologia do Hospital de Clínicas da Universidade Federal do Paraná, em Curitiba (PR), entre janeiro de 2010 e dezembro de 2011. O diagnóstico de DPOC foi baseado nos critérios da Global Initiative for Chronic Obstructive Lung Disease (GOLD)(9): limitação persistente e pro-gressiva ao fluxo aéreo; e resposta inflamatória crônica aumentada das vias aéreas e pulmões a partículas ou gases nocivos, evidenciada por relação VEF1/CVF pós-broncodilatador < 0,70 na espirometria.(9) Os critérios de inclusão foram ter DPOC relacionada ao tabagismo, ter realiza-do teste de função pulmonar com espirômetro (KoKo PFT; nSpire Health, Longmont, CO, EUA) e ter relação VEF1/CVF pós-broncodilatador < 0,70. Foram excluídos pacientes que não dispunham do resultado de qualquer um dos exames necessários, bem como aqueles que estavam tomando medicamentos que podem diminuir a massa magra e aqueles que tinham qualquer outra doença conhecida por afetar a composição corporal. O estudo foi aprovado pelo Comitê de Ética em Pesquisa Humana do Hospital de Clínicas, e todos os pacientes participantes assinaram um termo de consentimento livre e esclarecido.
Foram coletados dados clínicos, incluindo carga tabágica durante a vida, quantificada em anos-maço(10); história de exacerbações no ano an-terior; VEF1; escores obtidos na escala modificada de dispneia do Medical Research Council(11); escores obtidos no COPD Assessment Test, que quantifica o impacto geral da DPOC sobre o estado de saúde(12); e distância percorrida no teste de caminhada de seis minutos.(13)
Os pacientes foram classificados segundo o VEF1 em porcentagem do previsto (isto é, o grau de obstrução): ≥ 80% (leve); 50-79% (modera-do); 30-49% (grave); e < 30% (muito grave).(9) A gravidade (estádio clínico) da DPOC foi determinada de acordo com os critérios GOLD(9): VEF1 pós-broncodilatador; história de exacerbações no ano anterior; e sintomas tais como dispneia (medida pela escala modificada de dispneia do Medical Research Council ou pelo COPD Assessment Test). Com base nesses critérios, cada paciente foi categorizado como tendo DPOC no estádio GOLD I, II, III ou IV.(9) O prognóstico da DPOC foi avaliado pelo índice Body mass index, airflow Obstruction, Dyspnea, and Exercise capacity (BODE), estratificando-se os pacientes em quatro quartis, sendo o quarto o mais grave.(14)
O peso (em kg) foi medido, com os pacientes usando roupas leves, em balança eletrônica digital (Toledo do Brasil, São Bernardo do Campo, Brasil) com capacidade máxima de 200 kg e precisão de 50 g. A altura (m) foi medida com os pacientes em pé com as costas retas, os calca-nhares juntos e os braços ao lado do corpo. Com base no IMC, os pacientes foram classificados como tendo baixo peso (IMC < 22 kg/m2), peso normal (IMC ≥ 22 e < 27 kg/m2) ou sobrepeso/obesidade (IMC ≥ 27 kg/m2), de acordo com as categorias estabelecidas por Lipschitz et al.,(15) que são as mais adequadas para utilização em indivíduos de meia-idade e em idosos.
Para a avaliação da composição corporal, todos os pacientes realizaram dual-energy X-ray absorptiometry (DXA, absorciometria de dupla energia por raios X) em um scanner de corpo inteiro (Lunar Prodigy; GE Medical Systems, Madison, WI, EUA), utilizado em conjunto com o software enCORE 2002 (GE Medical Systems). O software fornece dados de massa magra corporal (massa óssea mais massa livre de gordura), massa magra livre de osso (massa magra menos massa livre de gordura), massa gorda e densidade mineral óssea.
Diagnóstico densitométrico de sarcopeniaOs critérios para o diagnóstico de sarcopenia por DXA foram inicialmente definidos por Baumgartner et al.(16) em um estudo com uma grande amostra de idosos no estado do Novo México. Porém, utilizando esses critérios, os autores constataram que a prevalência de sarcopenia era subdiagnosticada, provavelmente por causa das altas proporções de indivíduos com sobrepeso e obesidade na população estudada. Newman et al.(17) propuseram novos métodos para definir a sarcopenia, ajustando a altura e a massa magra apendicular (MMA) pela massa gorda, o que resultou em taxas de prevalência maiores do que as obtidas com os critérios anteriores.(17,18) Porém, esses autores constataram que os critérios anteriores continuavam superiores para diagnosticar a sarcopenia em pacientes com baixo peso. Em dois estudos recentes realizados no Brasil, Domiciano et al.(19) e Figueiredo et al.(20) compararam os dois conjuntos de critérios e concluíram que o ponto de corte de 22 kg/m2 para o IMC definiu a escolha dos critérios a serem utilizados para o diagnóstico densitométrico de sarcopenia.
Para o diagnóstico densitométrico de sarcopenia, foram utilizados ambos os sistemas de determinação da MMA: aquele que utiliza o índice de massa muscular esquelética (IMME), calculado como MMA (em kg) em relação ao quadrado da altura em metros (MMA/altura2), com pontos de corte de 7,26 kg/m2 e 5,45 kg/m2 para homens e mulheres, respectivamente, daqui por diante designado critérios de Baumgartner(16); e aquele que calcula a MMA (em kg) em relação à altura em metros e ajustada pela massa de gordura corporal total (em kg), daqui por diante designado critérios de Newman.(17) Os resíduos da regressão foram utilizados para identificar indivíduos com massa magra menor do que o valor previsto para uma determinada massa gorda (dado por uma equação derivada do modelo). Resíduos positivos indicariam indivíduos com relativa massa muscular, enquanto resíduos negativos indicariam indivíduos com sarcopenia. As equações derivadas dos modelos foram as seguintes:
para homens
MMA (em kg) = −28,15 + 27,49 × altura (em m) + 0,1106 × massa gorda (em kg)
para mulheres
MMA (em kg) = −19,78 + 20,00 × altura (em m) + 0,1554 × massa gorda (em kg)
O percentil 20 da distribuição dos resíduos foi utilizado como ponto de corte para o diagnóstico de sarcopenia, de acordo com a MMA ajustada pela massa gorda, como definido anteriormente.(17,18) Em nossa amostra, esse ponto de corte correspondeu a resíduos de −2,021 para homens e de −1,082 para mulheres. Para a realização do diagnóstico densitométrico de sarcopenia, aplicamos os critérios de Baumgartner para indiví-duos com IMC < 22 kg/m2 e os critérios de Newman para indivíduos com IMC ≥ 22 kg/m2.(21)
Análise estatísticaOs dados são apresentados em média ± dp, exceto onde especificado. Todas as análises estatísticas foram realizadas utilizando-se o software SPSS Statistics, versão 20.0 (IBM Corporation, Armonk, NY, EUA). Para as variáveis avaliadas, a normalidade da distribuição dos dados foi verificada com o teste de Kolmogorov-Smirnov. Para as comparações das variáveis quantitativas entre dois grupos, utilizou-se o teste t de Student para amostras independentes ou o teste não paramétrico de Mann-Whitney. Para as comparações entre três ou mais grupos, utilizaram-se one-way ANOVA e o teste da diferença mínima significativa para comparações múltiplas ou o teste não paramétrico de Kruskal-Wallis. Na análise estatística preliminar, utilizaram-se o teste exato de Fisher e o teste do qui-quadrado para avaliar a associação entre duas variáveis qualitativas. Valores de p < 0,05 foram considerados estatisticamente significativos.
Para a análise multivariada, utilizou-se regressão logística, tendo como variável resposta (dependente) a sarcopenia e como variáveis expli-cativas (independentes) a idade (≤ 67 ou > 67 anos), sexo feminino, estádio GOLD III ou IV, BODE nos quartis 3 ou 4 e tabagismo atual. Para cada variável, na presença da outra variável incluída no modelo, testou-se a hipótese nula de que a probabilidade de sarcopenia é igual para as duas classificações da variável (falta de associação entre a variável e a sarcopenia), versus a hipótese alternativa de diferentes probabilida-des. Calcularam-se os valores de p dos testes estatísticos e as razões de chances com seus respectivos intervalos de confiança de 95%.
RESULTADOSDos 96 pacientes avaliados, 5 foram excluídos por não terem realizado todos os testes necessários. Portanto, a amostra final foi composta por 91 pacientes (50 mulheres e 41 homens), com média de idade de 67,4 ± 8,7 anos e média de IMC de 25,8 ± 6,1 kg/m2. Vinte e cinco pacientes (27,4%) foram classificados como tendo peso normal, 28 (30,7%) como tendo baixo peso e 38 (41,7%) como tendo sobrepeso. A média percen-tual geral de massa de gordura corporal total foi de 32,3 ± 11,7% (37,8 ± 15,8% nas mulheres e 25,6 ± 7,1% nos homens; p = 0,000), e a média geral do IMME foi de 6,57 ± 1,1 (6,17 ± 5 para as mulheres e 7,05 ± 5,3 para os homens, p = 0,000). A carga tabágica média foi de 60 ± 41,4 anos-maço. Dezesseis pacientes (17,6%) eram fumantes no momento da inclusão no estudo.
Com base nos valores de VEF1, o grau de obstrução foi classificado como leve em 16 (17,6%) dos pacientes, moderado em 33 (36,3%), grave em 29 (31,9%) e muito grave em 13 (14,3%). Entre os 91 pacientes avaliados, a DPOC foi classificada como estádio GOLD I em 15 (16,5%), estádio GOLD II em 22 (24,2%), estádio GOLD III em 34 (37,4%) e estádio GOLD IV em 20 (22%). Na estratificação pelo índice BODE, 36 paci-entes (39,6%) estavam no quartil 1; 29 (31,9%) no quartil 2; 15 (16,5%) no quartil 3; e 11 (12,1%) no quartil 4. Constatou-se que o IMC não apresentou correlação significativa com VEF1 (p = 0,509), estádios GOLD (p = 0,114) ou quartis do BODE (p = 0,114). A sarcopenia foi diagnos-ticada em 36 pacientes (39,6%): 16 mulheres e 20 homens. Ao se estratificar esse resultado segundo o IMC, constatou-se que a sarcopenia estava presente em 5 (20,0%) dos 25 pacientes com peso normal, 23 (82,1%) dos 28 pacientes com baixo peso e 8 (21,1%) dos 38 pacientes com sobrepeso. A Figura 1 mostra o número de pacientes com sarcopenia, segundo o IMC, de acordo como o conjunto de critérios utilizado para o diagnóstico. Não houve diferenças significativas na prevalência de sarcopenia em relação a sexo (p = 0,276), idade (p = 0,309) ou carga tabágica (p = 0,464). A média do IMME foi de 5,86 ± 0,7 nos pacientes com sarcopenia (5,38 ± 0,42 nas mulheres e 6,25 ± 0,57 nos homens), contra 7,03 ± 1,0 naqueles sem sarcopenia (6,54 ± 0,89 nas mulheres e 7,81 ± 0,77 nos homens), sendo significativa a diferença entre os dois grupos (p < 0,001). Como se pode observar na Tabela 1, os pacientes com sarcopenia também apresentaram menor IMC (p < 0,001), menor percentual de gordura corporal total (p = 0.01) e menor massa magra total (p < 0,001)


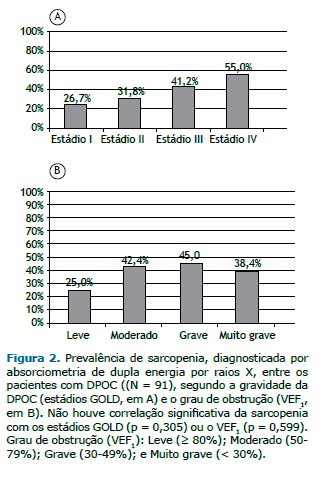
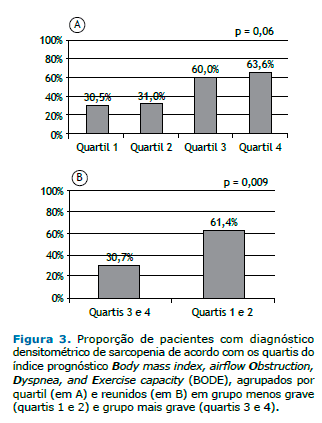
Não houve associação entre a prevalência de sarcopenia e a gravidade da DPOC, determinada pelo estádio GOLD e pelo VEF1 (Figuras 2A e 2B). Houve tendência de associação entre a prevalência de sarcopenia e os quartis do BODE (p = 0,06). Conforme ilustrado na Figura 3A, 11 pacientes (30,5%) estavam no quartil 1; 9 (31%) no quartil 2; 8 (60%) no quartil 3; e 9 (63,6%) no quartil 4. A Figura 3B mostra que a preva-lência de sarcopenia entre os pacientes nos quartis 3 ou 4 foi de 61,4%, significativamente maior do que os 30,7% observados entre aqueles nos quartis 1 ou 2 (p = 0,009; OR = 3,89; IC95%: 1,21-12,46). A análise multivariada mostrou que, independentemente de idade, sexo, status tabágico e estádio GOLD, o índice BODE estava significativamente associado à sarcopenia (Tabela 2).
 DISCUSSÃO
DISCUSSÃOEm nossa amostra de pacientes com DPOC, houve alta prevalência de sarcopenia diagnosticada por DXA, a qual se correlacionou com pior prognóstico. Em pacientes com DPOC, diversos fatores já se mostraram associados a pior prognóstico, maior número de internações e menor sobrevida; dentre os quais se destacam as alterações na composição corporal.(4) O baixo IMC já foi associado a exacerbações agudas, mortali-dade e perda de massa magra em pacientes com DPOC No presente estudo, confirmamos que a perda de massa magra corporal está associada a menor IMC em pacientes com DPOC. Porém, não foi constatada associação da massa magra com a gravidade da DPOC ou com pior prognósti-co, o que está de acordo com os achados de estudos recentes que demonstram que a redução da massa magra é um melhor preditor de morta-lidade em pacientes com DOPC do que o baixo IMC por si só, sendo que aquele reduz a sobrevida em até 50%.(22)
Em nosso estudo, a prevalência geral de sarcopenia foi de 39,6%, o que é consistente com a literatura sobre pacientes com DPOC, na qual a prevalência relatada é de 20-40%.(5,22) Cesari et al.(23) diagnosticaram sarcopenia em 30% dos pacientes com DOPC avaliados; quando os autores fizeram o ajuste pela massa gorda, essa taxa subiu para 42,5%, uma diferença atribuída principalmente à maior proporção de pacientes obesos diagnosticados. Ao aplicarmos os critérios de Baumgartner, diagnosticamos sarcopenia em 37 pacientes (40,6%), contra apenas 19 pacientes (21,0%) ao aplicarmos os critérios de Newman. Isso se deveu principalmente ao fato de que a média de IMC em nossa amostra foi menor do que a relatada nos outros estudos citados. Para os pacientes com sobrepeso e obesidade, o ajuste pela massa gorda (isto é, a aplicação dos critérios de Newman) foi especialmente importante, pois aumentou a prevalência de sarcopenia em 8% em relação à obtida ao se aplicarem os critérios de Baumgartner. Isso demonstra a importância de se fazer esse ajuste, o que pode evitar o subdiagnóstico.
A perda de massa magra corporal em pacientes com DPOC já se mostrou associada a menor IMC,(6) o que foi confirmado por nós. Porém, a sarcopenia também ocorre em aproximadamente 20% dos pacientes com peso normal, sobrepeso e obesidade. Um estudo mostrou que a dimi-nuição da massa muscular ocorre em 21% dos pacientes com peso normal com DPOC,(24) e outro mostrou que 10-15% dos pacientes com DPOC com IMC normal ou alto têm sarcopenia.(25)
O consumo de cigarros é um dos mecanismos envolvidos no aumento do catabolismo de proteínas na DOPC, como demonstrado em diversos estudos epidemiológicos de associação entre tabagismo e sarcopenia.(26) Embora haja controvérsias na literatura, um estudo demonstrou asso-ciação entre carga tabágica e perda de massa magra em pacientes com DPOC,(27) enquanto outros não demonstraram tal associação.(28,29) No presente estudo, não foi observada associação entre carga tabágica e sarcopenia. Uma possível explicação para essa achado é que pacientes com DPOC apresentam um estado inflamatório elevado e persistente, com altos níveis de TNF-α, o qual está envolvido na fisiopatologia da sarcopenia, independentemente da carga tabágica.(27)
Em nossa amostra de pacientes com DPOC, constatou-se que o diagnóstico de sarcopenia se correlacionou com menor IMC, a redução do percentual geral de gordura corporal, menor massa magra total e o IMME. Esses achados são consistentes com os de outros estudos sobre sarcopenia em pacientes com e sem DPOC,(30,31) já tendo sido demonstrada a ocorrência de uma redução anual de 1-2% na massa muscular ao longo de 50 anos em pacientes com DPOC.(32) A perda de massa magra corporal ocorre por vários mecanismos, tais como a morte de neurônios moto-res mediada pelo TNF-α, alterações hormonais, o estado nutricional e o aumento de fatores inflamatórios. Em nosso estudo, não foi observado aumento da prevalência de sarcopenia com o avanço da idade, o que está de acordo com os achados de outros estudos sobre DPOC.(33,34) Uma possível explicação para isso é que a gravidade da doença e os níveis elevados de citocinas inflamatórias, presentes desde o início da DPOC, contribuem para maior perda de massa magra, independentemente da idade.(35)
No presente estudo, nem o estádio GOLD nem o grau de obstrução (VEF1) se correlacionaram com o diagnóstico de sarcopenia. O número de pacientes estudados e a distribuição dos níveis de gravidade poderiam explicar essa falta de correlação. Contrariando nossos achados, Ischaki et al.(36) mostraram que, em pacientes com DPOC, quanto maior a perda de massa magra corporal, maior a gravidade da doença, quantificada pelos critérios GOLD e pelo VEF1. Porém, esses autores avaliaram a composição corporal utilizando bioimpedância elétrica, que é menos confi-ável do que DXA.(16) Em outro estudo recente, menor massa magra também se associou a pior VEF1, embora, novamente, a composição corporal tenha sido avaliada por bioimpedância elétrica.(37)
O índice BODE, um fator prognóstico, leva em consideração a capacidade de exercício, que pode afetar a massa magra. Observamos uma tendência a maior prevalência de sarcopenia entre os pacientes com DPOC com BODE nos quartis mais altos, e houve diferença estatisticamen-te significativa entre aqueles nos quartis mais baixos e aqueles nos quartis mais altos. Na análise multivariada, o índice BODE foi significativa-mente associado à sarcopenia, que foi mais prevalente entre os pacientes com pior prognóstico (nos quartis 3 ou 4). Até onde sabemos, não há outros estudos investigando essa associação. A redução da massa magra leva à intolerância ao exercício, que é descrita como fator essencial para o comprometimento da qualidade de vida, aumentando a frequência de exacerbações/internações hospitalares, e para o aumento da mortalidade.(4) Isso confirma que a sarcopenia está associada a pior prognóstico na DPOC.
Uma limitação de nosso estudo é que não foi avaliada a força muscular. Porém, diversos estudos demonstraram correlação entre a redução da massa magra corporal e a diminuição da força muscular.(38) Além disso, essa limitação foi provavelmente compensada pelo fato de termos aplicado o índice BODE, que leva em conta a capacidade de exercício.
Os resultados do presente estudo demonstram que a prevalência de sarcopenia, diagnosticada por DXA, que é atualmente considerada o mé-todo padrão ouro, foi alta em uma amostra de pacientes com DPOC. O nosso foi um estudo pioneiro no sentido de correlacionar a prevalência de sarcopenia com os quartis do índice BODE. O comprometimento e a perda de massa magra são comuns e preocupantes; como manifestações extrapulmonares da DPOC, causam redução da capacidade de exercício. Isso também pode resultar em perda ainda mais acentuada de massa muscular, iniciando assim um círculo vicioso. Portanto, o diagnóstico precoce de sarcopenia, por meio da análise da composição corporal, pode facilitar a implementação de intervenções voltadas à prevenção da deterioração da massa magra corporal e à melhora da qualidade de vida em pacientes com DPOC.
REFERÊNCIAS1. Eagan TM, Aukrust P, Ueland T, Hardie JA, Johannessen A, Mollnes TE, et al. Body composition and plasma levels of inflammatory biomarkers in COPD. Eur Respir J. 2010;36(5):1027-33. http://dx.doi.org/10.1183/09031936.00194209
2. Maltais F. Body composition in COPD: looking beyond BMI. Int J Tuberc Lung Dis. 2014;18(1):3-4. http://dx.doi.org/10.5588/ijtld.13.0868
3. Vilaró J, Ramirez-Sarmiento A, Martínez-Llorens JM, Mendoza T, Alvarez M, Sánchez-Cayado N, et al. Global muscle dysfunction as a risk factor of readmission to hospital due to COPD exacerbations. Respir Med. 2010;104(12):1896-902. http://dx.doi.org/10.1016/j.rmed.2010.05.001
4. Schols AM, Broekhuizen R, Weling-Scheepers CA, Wouters EF. Body composition and mortality in chronic obstructive pulmonary disease. Am J Clin Nutr. 2005;82(1): 53-9. http://dx.doi.org/10.1016/j.rmedu.2005.09.028
5. Marquis K, Debigaré R, Lacasse Y, LeBlanc P, Jobin J, Carrier G, Maltais F. Midthigh muscle cross-sectional area is a better predictor of mortality than body mass index in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2002;166(6):809-13. http://dx.doi.org/10.1164/rccm.2107031
6. Polkey MI, Moxham J. Attacking the disease spiral in chronic obstructive pulmonary disease. Clin Med. 2006;6(2):190-6. http://dx.doi.org/10.7861/clinmedicine.6-2-190
7. Santilli V, Bernetti A, Mangone M, Paoloni M. Clinical definition of sarcopenia. Clin Cases Miner Bone Metab. 2014;11(3):177-80. http://dx.doi.org/10.11138/ccmbm/2014.11.3.177
8. Franssen FM, Sauerwein HP, Rutten EP, Wouters EF, Schols AM. Whole-body resting and exercise-induced lipolysis in sarcopenic [corrected] patients with COPD. Eur Respir J. 2008;32(6):1466-71. Erratum in: Eur Respir J. 2009;33(4):947. http://dx.doi.org/10.1183/09031936.00014008
9. Global Initiative for Chronic Obstructive Lung Disease [homepage on the Internet]. Bethesda: GOLD [cited 2015 Feb 27]. Global Strategy for the Diagnosis,
Management, and Prevention of Chronic Obstructive Lung Disease (Revised 2011). [Adobe Acrobat document, 90p.]. Available from: http://www.goldcopd.org/uploads/users/files/GOLD_Report_2011_Feb21.pdf
10. Prignot J. Quantification and chemical markers of tobacco-exposure. Eur J Respir Dis. 1987;70(1):1-7.
11. Ferris BG. Epidemiology Standardization Project (American Thoracic Society). Am Rev Respir Dis. 1978;118(6 Pt 2):1-120.
12. Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD Assessment Test. Eur Respir J. 2009;34(3):648-54. http://dx.doi.org/10.1183/09031936.00102509
13. BALKE B. A SIMPLE FIELD TEST FOR THE ASSESSMENT OF PHYSICAL FITNESS. REP 63-6. Rep Civ Aeromed Res Inst US. 1963 Apr:1-8.
14. Celli BR, Cote CG, Marin JM, Casanova C, Montes de Oca M, Mendez RA, et al. The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med. 2004;350(10):1005-12. http://dx.doi.org/10.1056/NEJMoa021322
15. Lipschitz DA. Screening for nutritional status in the elderly. Prim Care. 1994;21(1):55-67.
16. Baumgartner RN, Koehler KM, Gallagher D, Romero L, Heymsfield SB, Ross RR, et al. Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol. 1998;147(8):755-63. http://dx.doi.org/10.1093/oxfordjournals.aje.a009520
17. Newman AB, Kupelian V, Visser M, Simonsick E, Goodpaster B, Nevitt M, et al. Sarcopenia: alternative definitions and associations with lower extremity function. J Am Geriatr Soc. 2003;51(11):1602-9. http://dx.doi.org/10.1046/j.1532-5415.2003.51534.x
18. Delmonico MJ, Harris TB, Lee JS, Visser M, Nevitt M, Kritchevsky SB, et al. Health, Aging and Body Composition Study. Alternative definitions of sarcopenia, lower extremity performance, and functional impairment with aging in older men and women. J Am Geriatr Soc. 2007;55(5):769-74. http://dx.doi.org/10.1111/j.1532-5415.2007.01140.x
19. Domiciano DS, Figueiredo CP, Lopes JB, Caparbo VF, Takayama L, Menezes PR, et al. Discriminating sarcopenia in community-dwelling older women with high frequency of overweight/obesity: the São Paulo Ageing & Health Study (SPAH). Osteoporos Int. 2013;24(2):595-603. http://dx.doi.org/10.1007/s00198-012-2002-1
20. Figueiredo CP, Domiciano DS, Lopes JB, Caparbo VF, Scazufca M, Bonfá E, Pereira RM. Prevalence of sarcopenia and associated risk factors by two diagnostic criteria in community-dwelling older men: the São Paulo Ageing & Health Study (SPAH). Osteoporos Int. 2014;25(2):589-96. http://dx.doi.org/10.1007/s00198-013-2455-x
21. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-23. http://dx.doi.org/10.1093/ageing/afq034
22. Wagner PD. Possible mechanisms underlying the development of cachexia in COPD. Eur Respir J. 2008;31(3):492-501. http://dx.doi.org/10.1183/09031936.00074807
23. Cesari M, Pedone C, Chiurco D, Cortese L, Conte ME, Scarlata S, et al. Physical performance, sarcopenia and respiratory function in older patients with chronic obstructive pulmonary disease. Age Ageing. 2012;41(2):237-41. http://dx.doi.org/10.1093/ageing/afr167
24. Bolton CE, Ionescu AA, Shiels KM, Pettit RJ, Edwards PH, Stone MD, et al. Associated loss of fat-free mass and bone mineral density in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2004;170(12):1286-93. http://dx.doi.org/10.1164/rccm.200406-754OC
25. Vestbo J, Prescott E, Almdal T, Dahl M, Nordestgaard BG, Andersen T, et al. Body mass, fat-free body mass, and prognosis in patients with chronic obstructive pulmonary disease from a random population sample: findings from the Copenhagen City Heart Study. Am J Respir Crit Care Med. 2006;173(1):79-83. http://dx.doi.org/10.1164/rccm.200506-969oc
26. Rom O, Kaisari S, Aizenbud D, Reznick AZ. Sarcopenia and smoking: a possible cellular model of cigarette smoke effects on muscle protein breakdown. Ann N Y Acad Sci. 2012;1259:47-53. http://dx.doi.org/10.1111/j.1749-6632.2012.06532.x
27. Eagan TM, Gabazza EC, D'Alessandro-Gabazza C, Gil-Bernabe P, Aoki S, Hardie JA, et al. TNF-α is associated with loss of lean body mass only in already cachectic COPD patients. Respir Res. 2012;13:48. http://dx.doi.org/10.1186/1465-9921-13-48
28. Tanni SE, Pelegrino NR, Angeleli AY, Correa C, Godoy I. Smoking status and tumor necrosis factor-alpha mediated systemic inflammation in COPD patients. J Inflamm (Lond). 2010;7:29. http://dx.doi.org/10.1186/1476-9255-7-29
29. Iwaniec UT, Fung YK, Cullen DM, Akhter MP, Haven MC, Schmid M. Effects of nicotine on bone and calciotropic hormones in growing female rats. Calcif Tissue Int. 2000;67(1):68-74. http://dx.doi.org/10.1007/s00223001099
30. Rutten EP, Calverley PM, Casaburi R, Agusti A, Bakke P, Celli B, et al. Changes in body composition in patients with chronic obstructive pulmonary disease: do they influence patient-related outcomes? Ann Nutr Metab. 2013;63(3):239-47. http://dx.doi.org/10.1159/000353211
31. Verhage TL, Heijdra Y, Molema J, Vercoulen J, Dekhuijzen R. Associations of muscle depletion with health status. Another gender difference in COPD? Clin Nutr. 2011;30(3):332-8. http://dx.doi.org/10.1016/j.clnu.2010.09.013
32. Kim TN, Choi KM. Sarcopenia: definition, epidemiology, and pathophysiology. J Bone Metab. 2013;20(1):1-10. http://dx.doi.org/10.11005/jbm.2013.20.1.1
33. Agustí A, Barberà JA, Wouters EF, Peinado VI, Jeffery PK. Lungs, bone marrow, and adipose tissue. A network approach to the pathobiology of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2013;188(12):1396-406. http://dx.doi.org/10.1164/rccm.201308-1404PP
34. Hopkinson NS, Tennant RC, Dayer MJ, Swallow EB, Hansel TT, Moxham J, et al. A prospective study of decline in fat free mass and skeletal muscle strength in chronic obstructive pulmonary disease. Respir Res. 2007;8:25. http://dx.doi.org/10.1186/1465-9921-8-25
35. Ferrucci L, Penninx BW, Volpato S, Harris TB, Bandeen-Roche K, Balfour J, et al. Change in muscle strength explains accelerated decline of physical function in older women with high interleukin-6 serum levels. J Am Geriatr Soc. 2002;50(12):1947-54. http://dx.doi.org/10.1046/j.1532-5415.2002.50605.x
36. Ischaki E, Papatheodorou G, Gaki E, Papa I, Koulouris N, Loukides S. Body mass and fat-free mass indices in COPD: relation with variables expressing disease severity. Chest. 2007;132(1):164-9. http://dx.doi.org/10.1378/chest.06-2789
37. Abbatecola AM, Fumagalli A, Spazzafumo L, Betti V, Misuraca C, Corsonello A, et al. Body composition markers in older persons with COPD. Age Ageing. 2014;43(4):548-53. http://dx.doi.org/10.1093/ageing/aft196
38. Hansen RD, Raja C, Aslani A, Smith RC, Allen BJ. Determination of skeletal muscle and fat-free mass by nuclear and dual-energy x-ray absorptiometry methods in men and women aged 51-84 y (1-3). Am J Clin Nutr. 1999;70(2):228-33.






