To the Editor:A 20-year old woman was referred to the Department of Thoracic Surgery of Hospital Sírio Libanês as a candidate for thymectomy. She had a two-year history of myasthenia gravis, presenting with diplopia, ptosis, and mild generalized weakness (stage IIA according to the Osserman classification).
The physical examination was unremarkable. Preoperative procedures included chest radiograph, CT of the chest, pulmonary function tests, and a complete neurological work-up. Blood tests were positive for acetylcholine receptor antibodies. The CT findings excluded the possibility of thymoma. The patient was deemed a good candidate for thymectomy by thoracoscopic robot-assisted surgery.
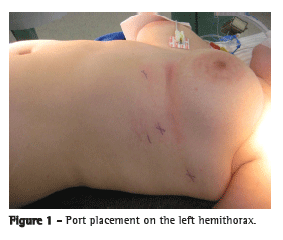
The patient was submitted to general and epidural anesthesia, a double-lumen endotracheal tube was placed, and she was positioned in a left-sided 30° elevated lateral decubitus. The camera port was placed in the fourth intercostal space at the anterior axillary line, and the instrument ports were placed in the third intercostal space at the mid-axillary line and in the fifth intercostal space at the midclavicular line (Figure 1). A three-dimensional 0° camera was introduced for the complete visualization of the pleural space, and the two arms of the robotic surgical system (da Vinci®; Intuitive Surgical, Inc., Menlo Park, CA, USA) were attached. For the left arm, an EndoWrist® instrument (Intuitive Surgical, Inc.) was used, whereas an electrocautery unit was used for the right arm. The left arm was mainly used to grasp the thymus, and the dissection was performed with the right arm.
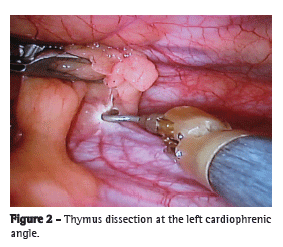
The dissection began inferiorly at the left cardiophrenic angle along the left phrenic nerve (Figure 2). The thymus gland was separated from the retrosternal area, and the left inferior horn of the thymus was isolated and dissected from the pericardium.
As the dissection proceeded, the innominate vein was identified, and the thymic veins were clipped and divided. The cardiophrenic angle was then dissected on the right side, and both upper thymic poles were isolated. The dissection finished at the lower right side, with identification of the right phrenic nerve and subsequent exploration of the pleura.
The thymus gland and the anterior mediastinal/cervical fat tissue were resected radically and removed through the trocar port. After hemostasis had been achieved, a 28F drainage chest tube was inserted into the pleural cavity through the port of the fifth intercostal space, the lung was reinflated, and the other wounds were closed in a standard fashion. The total operative time was 120 min, including the robotic docking, thymus resection, and skin closure.
The postoperative period was uneventful, the chest tube was removed 48 h after surgery, and the patient was discharged 72 h after surgery. The final histology revealed thymic hyperplasia, with no ectopic foci in the resected surrounding tissue.
After a three-month follow-up period, the patient was clinically well, and a reduction in the prednisone dose was possible, although the cholinesterase inhibitors were kept at the same dosage. Although she had pain at the incision site for approximately one month, she returned to work 15 days after surgery and was able to resume light sports one month after hospital discharge.
The first thymectomy by robot-assisted surgery for the treatment of a small thymoma was performed in 2001,(1) and, throughout that decade, many reports confirmed the advantages of this new technique for thymus resection. Myasthenia gravis is an autoimmune disease that affects neuromuscular transmission and results in chronic weakness and various levels of fatigue in striated muscles. The pathophysiology is related to the presence of acetylcholine receptors generated in the thymus, which ultimately lead to the symptoms.
Thymectomy is a widely accepted therapeutic option as an adjunct to clinical treatment, yielding positive results in terms of improvement and remission of symptoms. In this setting, numerous thymectomy techniques, of varying degrees of invasiveness, have been proposed, generating controversy regarding the best surgical approach for myasthenia gravis.(2-5) The optimal timing for thymectomy has yet to be established; however, it is probably best performed when patients still have minimal bulbar and respiratory symptoms. If the clinical status allows, it is also helpful to lower the dosage of corticoids. Some authors have suggested that the response is better if thymectomy is performed as soon as the disease is diagnosed.
Analyzing the literature, we have found that transcervical thymectomy is limited to the removal of the intracapsular portion of the lobes. Therefore, this is associated with a higher amount of residual thymus and, as a consequence, with a higher operation rate without the remission of symptoms due to ectopic foci.(1) One means of overcoming this problem is to perform extended transcervical thymectomy using a sternal retractor under the manubrium, although that does not allow the surgeon to remove the layers of the pleura.(2) Therefore, combined transsternal and cervical thymectomy consists of the en-bloc removal of all thymus in the neck and mediastinum. It is the benchmark operation, which other approaches are to be compared with.(2)
Recent advances in video-assisted thoracoscopic surgery (VATS) and minimally invasive surgical techniques have made thymectomy possible. These approaches have reduced morbidity, mortality, infection rates, postoperative hospital stays, and the use of pain medication.(3)
The da Vinci® surgical system consists of an input device (surgeon console) and an output device (patient-side manipulator) with two or three instrument arms and one camera arm. The surgeon operates the console, which translates the position and the motion of the hands over highly sensitive motion sensors to the end of the instruments at a remote location. The instrument arms have seven degrees of freedom, promoting motion scaling and tremor filtering. In addition, the EndoWrist® surgical instrument has a 360° range of motion, improving maneuverability around anatomic structures.(4)
The disadvantages of this technique include loss of tactile sense, limited instrumentation compared with the VATS technique, and higher costs. The outcomes are comparable to those obtained with VATS, such as shorter hospital stays and less pain.(5)
We could safely visualize and dissect the upper lobes of the thymus, with a radical and complete resection, including all of the fat from the pericardium down to the diaphragm. We opted for a left-side approach, because it allows easier access to the aortopulmonary window, which is a common location of ectopic thymic tissue.(6) In addition, a left-side approach makes it easier to identify the phrenic nerve, which is mandatory in this population of patients.
We believe that, with the robotic approach, radical thymectomy can be performed safely, and this might lower the threshold for neurologists to recommend surgery for their patients with myasthenia gravis.
Rodrigo Afonso da Silva Sardenberg
Thoracic Surgeon,
Hospital Sírio Libanês and
Hospital Albert Einstein, São Paulo, Brazil
Ricardo Zugaib Abadalla
General Surgeon,
Hospital Sírio Libanês, São Paulo, Brazil
Igor Renato Louro Bruno Abreu
Thoracic Surgeon,
Hospital Sírio Libanês, São Paulo, Brazil
Eli Faria Evaristo
Neurologist,
Hospital Sírio Libanês and
Hospital das Clínicas,
University of São Paulo School of Medicine, São Paulo, Brazil
Riad Naim Younes
Associate Professor,
Department of Surgery,
University of São Paulo School of Medicine, São Paulo, Brazil.
Director of the Advanced Center for Thoracic Diseases, Hospital Sírio Libanês, São Paulo, BrazilReferences1. Jaretzki A 3rd. Thymectomy for myasthenia gravis: analysis of controversies--patient management. Neurologist. 2003;9(2):77-92.
2. Cooper JD, Al-Jilaihawa AN, Pearson FG, Humphrey JG, Humphrey HE. An improved technique to facilitate transcervical thymectomy for myasthenia gravis. Ann Thorac Surg. 1988;45(3):242-7.
3. Mack MJ, Landreneau RJ, Yim AP, Hazelrigg SR, Scruggs GR. Results of video-assisted thymectomy in patients with myasthenia gravis. J Thorac Cardiovasc Surg. 1996;112(5):1352-9; discussion 1359-60.
4. Hashizume M, Konishi K, Tsutsumi N, Yamaguchi S, Shimabukuro R. A new era of robotic surgery assisted by a computer-enhanced surgical system. Surgery. 2002;131(1 Suppl):S330-3.
5. Demmy TL, James TA, Swanson SJ, McKenna RJ Jr, D'Amico TA. Troubleshooting video-assisted thoracic surgery lobectomy. Ann Thorac Surg. 2005;79(5):1744-52; discussion 1753.
6. Rückert JC, Czyzewski D, Pest S, Müller JM. Radicality of thoracoscopic thymectomy--an anatomical study. Eur J Cardiothorac Surg. 2000;18(6):735-6.





