ABSTRACT
Objective: To evaluate the effects of immunization with a DNA-hsp65 vaccine in an experimental model of pulmonary fibrosis. Methods: A total of 120 male C57BL/6 mice were distributed into four groups: SS, injected with saline (placebo) and then receiving intratracheal (IT) instillation of saline; SB, injected with saline (placebo) and then receiving IT instillation of bleomycin; PB, treated with plasmid only, without bacterial genome, and then receiving IT instillation of bleomycin; and BB, treated with the vaccine and then receiving IT instillation of bleomycin. Bleomycin was instilled 15 days after the last immunization, and the animals were killed six weeks thereafter. The left and right lungs were removed, the former for morphological analysis and the latter for hydroxyproline measurements. Results: The proportion of deaths within the first 48 h after the IT instillation (deaths attributed to the surgical procedure) was higher in the SB group than in the SS group (57.7% vs. 11.1%). The mean area of pulmonary interstitial septa was greater in the SB and PB groups (53.1 ± 8.6% and 53.6 ± 9.3%, respectively) than in the SS and BB groups (32.9 ± 2.7% and 34.3 ± 6.1%, respectively). The mean area of interstitial septa stained by picrosirius was greater in the SB, PB and BB groups than in the SS group (8.2 ± 4.9%, 7.2 ± 4.2% and 6.6 ± 4.1%, respectively, vs. 2.0 ± 1.4%). The total hydroxyproline content in the lung was significantly lower in the SS group (104.9 ± 20.9 pg/lung) than in the other groups (SB: 160.4 ± 47.8 pg/lung; PB: 170.0 ± 72.0 pg/lung; and BB: 162.5 ± 39.7 pg/lung). Conclusions: Immunization with the DNA-hsp65 vaccine reduced the deposition of noncollagen matrix in a model of bleomycin-induced lung lesion.
Keywords:
Pulmonary fibrosis; Bleomycin; Collagen.
RESUMO
Objetivo: Avaliar a influência do biofármaco DNA-hsp65 em um modelo de distúrbio fibrosante pulmonar experimental. Métodos: Foram estudados 120 camundongos machos C57BL/6, divididos em quatro grupos: grupo SS, animais tratados com salina (placebo) e injetados com salina intratraqueal (IT); grupo SB, tratados com salina (placebo) e injetados com bleomicina IT; grupo PB, tratados com plasmídeo, sem gene bacteriano, e injetados com bleomicina IT; e grupo BB, tratados com DNA-hsp65 e injetados com bleomicina IT. A bleomicina foi injetada 15 dias após a última imunização, e os animais sacrificados seis semanas após o uso da droga IT. O pulmão esquerdo retirado foi utilizado para análise morfológica, e o pulmão direito para dosagens de hidroxiprolina. Resultados: A proporção de camundongos que apresentaram morte não-programada depois de 48 h da injeção IT foi maior no grupo SB em comparação ao grupo SS (57,7% vs. 11,1%). A área percentual média de interstício septal foi maior nos grupos SB e PB (53,1 ± 8,6% e 53,6 ± 9,3%, respectivamente) em comparação aos grupos SS e BB (32,9 ± 2,7% e 34,3 ± 6,1%, respectivamente). Os grupos SB, PB e BB mostraram aumentos nos valores médios da área de interstício septal corada por picrosirius em comparação ao grupo SS (SS: 2,0 ± 1,4%; SB: 8,2 ± 4,9%; PB: 7,2 ± 4,2%; e BB: 6,6 ± 4,1%). O conteúdo pulmonar de hidroxiprolina no grupo SS foi inferior ao dos demais grupos (SS: 104,9 ± 20,9 pg/pulmão; SB: 160,4 ± 47,8 pg/pulmão; PB: 170,0 ± 72,0 pg/pulmão; e BB: 162,5 ± 39,7 pg/pulmão). Conclusões: A imunização com o biofármaco DNA-hsp65 interferiu na deposição de matriz não-colágena em um modelo de lesão pulmonar induzida por bleomicina.
Palavras-chave:
Fibrose pulmonar; Bleomicina; Colágeno.
IntroductionPulmonary interstitial diseases constitute a heterogeneous group of illnesses in which varying degrees of inflammation and fibrosis can be observed.(1) Idiopathic pulmonary fibrosis (IPF) is a distinct type of chronic fibrosing interstitial pneumonia that is progressive, and the outcome is typically fatal.(1,2) It constitutes the paradigm of interstitial pulmonary diseases, both for its frequency and for its severity. Currently, there are no truly efficacious options for treating this disease.(2,3)
Among the many factors that can influence the scarring process and the final degree of fibrosis is the type of immune-inflammatory response involved. Among other factors, some studies indicate that T lymphocytes play an important role in the physiopathological process of IPF.(4,5) It is known that, in the interstitium, responses involving T helper 2 (Th2) lymphocytes predominate in patients with this disease.(6,7) In pulmonary lesions caused by this diseased there are mastocytes and eosinophils, as well as increased quantities of interleukin (IL)-4 and IL-13. The finding of reduction of the expression of the Th1 cytokine interferon-gamma (IFN-γ), which can activate cellular mechanisms of antigen removal and restoration of healthy tissue, can have implications in the establishment of pulmonary fibrotic lesions. A study involving lung biopsies of 10 patients with IPF characterized the types of cellular infiltrates using immunohistochemical techniques.(8) The majority of mononuclear cells infiltrating the tissue (> 50%) were stained for IL-4 and IL-5, whereas < 7% were stained for IFN-γ. In addition, there is accumulating evidence that the macrophages of patients with pulmonary fibrosis present increased expression of IL-10 and IL-13, cytokines equally related to Th2 inflammatory responses.(9,10) This set of observations suggests that the predominance of a Th2 immune-inflammatory response in pulmonary fibrosis over Th1 responses can contribute to the development of pulmonary fibrosis.
Recently, a vaccine based on the technology of the recombinant DNA, which uses the 65-kDa heat shock protein (hsp65) as the antigen, was developed in Brazil and has shown promising results in the prevention and treatment of tuberculosis in rodents.(11-15) Studies have shown that, in animals infected with the tuberculosis bacillus and exposed to this antigen, the initial immunological response is altered, promoting the expression of Th1 cytokines, with a consequent increase in IFN-γ and decrease in the numbers of bacterial colony-forming units. Due to these findings, we raised the hypothesis that immunization of animals with the DNA-hsp65 vaccine promotes protection against inflammatory and fibrosing alterations induced by the intratracheal (IT) instillation of bleomycin in mice.
MethodsA total of 120 male C57BL/6 mice, eight to ten weeks of age and free of specific germs, were studied. Throughout the experimental period, the animals were housed on ventilated shelves, in facilities appropriate for the maintenance of germ-free animals, at the Central Animal Facility of the Department of Clinical Medicine of the Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (FMRP-USP, University of São Paulo at Ribeirão Preto School of Medicine). The study was approved by the FMRP-USP Committee for Ethics in Animal Experimentation.
The mice were randomized into four groups:
1) SS group: injected with saline (placebo) and then receiving intratracheal (IT) instillation of saline
2) SB group: injected with saline (placebo) and then receiving IT instillation of bleomycin
3) PB group: animals treated with the DNA-hsp65 vaccine without bacterial genome (plasmid only), and then receiving IT instillation of bleomycin
4) BB group: animals treated with the DNA-hsp65 vaccine and then receiving IT instillation of bleomycin
The DNA-hsp65 vaccine was developed by cloning a DNA fragment of the genome that encodes the hsp65 gene of M. leprae in the pcDNA3 plasmid, as previously described.(15) The animals were immunized with injections into the quadriceps, at a dose of 100 µg and a volume of 50 µl per dose.
Corresponding volumes of plasmid only or saline were administered in the control groups. The immunization schedule encompassed four doses administered with a two-week interval between each dose.
In order to induce lung injury, bleomycin sulfate (Blenoxane®; Bristol Myers-Squibb, São Paulo, Brazil) was administered IT at 0.15 U for each 30 g of weight, two weeks after the last dose of vaccine, plasmid or saline. On this occasion, the animals were initially anesthetized with 2.5% tribromoethanol (Sigma-Aldrich, St. Louis, MO, USA) at 1 mL for each 100 g of weight, injected intraperitoneally. The animals were then immobilized with adhesive tape on a wooden plank, in the supine position and with the head hyperextended. A median cervical incision of 1-1.5 cm was made, with dilatation of the structures and exposure of the trachea. The corresponding doses of bleomycin diluted in distilled water were aspirated, and the final volume was brought up to 0.1 mL, using a syringe and a thin hypodermic needle, aiming to promote better distribution of the solution in the airways. Corresponding volumes of saline were similarly instilled in the control groups. At the end of the instillation, the cervical incision was sutured with fine cotton thread.
The animals were killed six weeks after IT instillations. Prior to sacrifice, the mice were anesthetized with intraperitoneal injections of 2.5% tribromoethanol and a cervical incision was made. After exposure, the trachea was sectioned and a small-diameter plastic catheter was introduced. An abdominal incision was made, and the descending aorta was sectioned while the animal was still alive and breathing spontaneously. The mice were then killed by exsanguination, after which the chest was opened and the cardiopulmonary block was removed for dissection. Using a fine cotton thread, the right main bronchi was ligated and sectioned. When freed, the right lung was weighed and stored at −70 °C for later biochemical analysis. Buffered 10% formalin was infused through the thin plastic catheter at a pressure of 20 cmH2O. The extremity of the trachea was then ligated with cotton thread and the plastic catheter was removed, after which the formalin-inflated lung was fixed by immersion in a receptacle also containing formalin. Left lungs were stored in formalin for later histological studies.
After standard processing, sagittal sections of left lungs were submitted to histological examination. The fixed lungs were stained with hematoxylin-eosin (H&E) and picrosirius.(16) The H&E-stained sections were mounted on slides and morphometrically analyzed by a pulmonary pathologist specialized in histological alterations of the septal interstitium. The findings are expressed as percentages of the total area of the field. In each slide, 10 randomly chosen fields were analyzed per pulmonary region, at high magnification (×400). A single value for alveolar septal thickness was established for each lung, through the calculation of the mean values found for the 10 fields. This number reflects the degree of alveolar collapse and of the established repair process, higher numbers indicating more pronounced lung injury.
For the morphometric analysis of collagen, the picrosirius technique, which specifically stains the collagen, was used, in conjunction with a digital system of imaging analysis (Leica Qwin; Leitz, Wetzlar, Germany) connected to an optical microscope (Leica DMR; Leitz) and to a personal computer. The area occupied by the fibers was determined through digital densitometry recognition and is expressed as a percentage of the total area of the field.
Lung hydroxyproline content was measured using the right lungs in their totality, through the colorimetric method devised by Rodjkind & Gonzáles and modified by Frankel et al.(17) The results are expressed as the total quantity of hydroxyproline per lung.
The results obtained are expressed as means and standard deviations. The results as shown as box plots. The analysis of the unexpected death rate was conducted for two periods: ≤ 48 h after the IT instillations ("early mortality"); and > 48 h after the IT instillations ("late mortality"). This distinction was made in order to distinguish deaths related to the surgical procedure per se from those related to the establishment of lung injury due to chemical insult. The mortality rates in the groups were compared using paired Fisher's exact test, and the p values were submitted to Bonferroni correction for multiple comparisons. The comparisons of the variables related to morphometry and lung hydroxyproline content were carried out using one-way analysis of variance, followed by Student-Newman-Keuls post-test, when indicated. The software GraphPad InStat (GraphPad Software Inc, San Diego, CA, USA) was used in those calculations. In all comparisons, the level of significance required in order to reject the null hypothesis was set at 5% (p < 0.05).
ResultsThe numbers of animals designated for each group are listed in Table 1, together with the early and late death rates. Although the percentage of early deaths was equal among the groups, the late death rate was significantly higher in the SB group than in the SS group. Although the percentage of late deaths was lower in the BB group than in the SB and PB groups, the difference was not statistically significant.
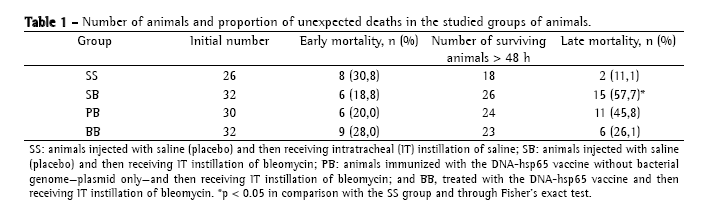
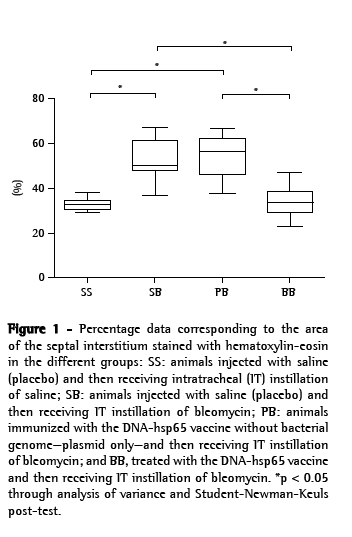
The mean percentage of the interstitial septal matrix stained with H&E was significantly lower in the BB group than in the SB and PB groups, although it was comparable to that found for the SS group (BB: 34.3 ± 6.1%; SS: 32.9 ± 2.7%; SB : 53.1 ± 8.6%; PB: 53.6 ± 9.3%; Figure 1). The mean percentage of the interstitial septal matrix stained with picrosirius was significantly lower in the SS Group than in the other groups (SS: 2.0 ± 1.4%; SB: 8.2 ± 4.9%; PB: 7.2 ± 4.2%; BB: 6.6 ± 4.1%; Figure 2).
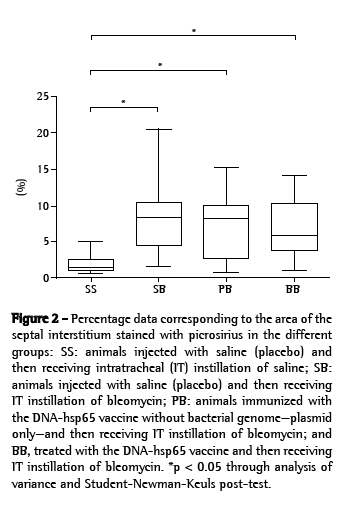
The total lung hydroxyproline content was significantly lower in the SS group than in the other groups (SS: 104.9 ± 20.9 pg/lung; SB: 160.4 ± 47.8 pg/lung; PB: 170.0 ± 72.0 pg/lung; BB: 162.5 ± 39.7 pg/lung; Figure 3). Figure 4 illustrates the histological aspects of the studied groups. In those images, the histological damage induced by bleomycin can clearly be seen, as can the degree of attenuation of these alterations in the BB-group lung stained with H&E.
 Discussion
DiscussionThere are a great number of non-infectious and non-neoplastic processes that present progressive, inflammatory fibrotic infiltration of the lungs and distortion of the parenchyma. Such processes are commonly associated with clinical severity and poor prognostic profiles. Although corticosteroids and immunosuppressants are effective in many situations, their frequent use is associated with undesirable side effects and even death. Moreover, IPF currently constitutes a private therapeutic challenge. For many authors, there is no clinical treatment that has proved effective against this disease.(18) These facts justify the great volume of experimental and clinical investigations involving potential therapeutic agents for these diseases. However, traditional approaches to the problem typically involve the use of pharmacological agents with local effects on only one or only a few of the points considered potentially important in the morbid process. Investigations carried out with various antioxidant agents constitute an instance of such.(19,20)
The DNA-hsp65 vaccine was initially developed in order to promote protection against infections caused by M. tuberculosis. However, there is mounting evidence that this vaccine, in addition to protecting animals against tuberculosis infection, can act as a therapeutic agent when the disease is already established. The vaccine was obtained by cloning the gene that encodes hsp65 in plasmids. To that end, plasmids expressing the promoter of the immediate early gene of the cytomegalovirus, or another originating from hydroxymethylglutaryl coenzyme A reductase in mice, were employed, with similar results.(15)
Many studies show that the DNA-hsp65 vaccine is an immunomodulatory agent and a powerful stimulant of the cellular immune response with efficacy superior even to that of the bacillus Calmette-Guérin.(11-15,21,22) Its intramuscular administration prior to M. tuberculosis infection leads to an elevation of the number of hsp65-reactive T cells. This increase occurs simultaneous to a drop in the numbers of CD4+/CD8− cells, as well as of CD4−/CD8+ cells. Immunization with the vaccine also leads to increased numbers of splenic T cells that express the CD44-hi phenotype, a marker associated with Th1 type responses. Regardless of their phenotype, hsp65-reactive cells from immunized animals produce elevated levels of IFN-γ. Conversely, the lymphocytes of unvaccinated animals respond to hsp65 stimuli in vitro, producing predominantly IL-4. The administration of the vaccine eight weeks after inoculation with the tuberculosis bacilli, in addition to reducing the number of germs at the tissue level, also leads to similar immunological alterations, that is, deviation of a Th2 immune response to a Th1 pattern. It is of note that animals not infected with the tuberculosis bacillus and treated only with the vaccine also present a significant elevation in the number of lymphocytes expressing IFN-γ in comparison with unvaccinated control animals.(21)
These findings support the application of the new agent in clinical scenarios in which increasing the cellular immune response could be beneficial. In a pilot study, the intratumoral application of the DNA-hsp65 vaccine in patients with advanced phase head or neck carcinoma was well tolerated and was, in some cases, associated with a reduction of tumor volume.(23)
As previously mentioned, studies conducted principally with material from lung biopsies and bronchoalveolar lavage fluid obtained from patients with IPF suggest that the excessive secretion of various cytokines classified as Th2 can contribute to the genesis of the disease. Conversely, experimental data show that administration of the Th1 IFN-γ cytokine can have a protective effect against the establishment of pulmonary fibrotic processes, although the results of studies involving the use of this substance in human beings with IPF have been disappointing.(24,25)
We developed the present study based on the hypothesis that immune stimulation with the DNA-hsp65 vaccine could be protective against the establishment of pulmonary fibrosis. The experimental model used was bleomycin-induced lung injury. Although the model cited does not reproduce exactly the histological pattern of IPF, it has been widely used as a screening tool for the evaluation of the efficacy of drugs with therapeutic potential for pulmonary fibrotic processes. In addition, the doses of the DNA-hsp65 employed in this study were based on previous studies in which the immunization schedule proved effective in stimulating murine immunity and providing protection against tuberculosis infection.(22)
Since the beginning of the project, we have observed elevated mortality rates among mice in the first hours following manipulation for the IT instillation of substances. This phenomenon occurred to the same degree in all groups and probably reflects the development of respiratory insufficiency and other complications related to the surgical manipulation of small-diameter tracheas.
Therefore, we chose to analyze only the unexpected deaths of animals surviving the first 48 h. Although the proportion of unexpected deaths in the BB group was statistically similar to that seen in the other groups, it was substantially lower than that seen in the SB group (26.1% vs. 57.7%). From our point of view, this suggests that the DNA-hsp65 vaccine can bring some degree of protection against lung aggression induced by the IT instillation of bleomycin. Findings referring to the measurements of the septal interstitium evaluated through morphometry in H&E-stained material point in the same direction. These measurements provide a yardstick of the global involvement and of the degree of the pulmonary septal interstitium response to chemical injury. In this aspect, the area of the septal interstitium of the BB group was significantly inferior to that of the SB and PB groups and comparable to that of the SS group.
The picrosirius staining technique stains specifically collagen fibers. Likewise, the tissue values of the amino acid hydroxyproline reflect the degree of deposition of collagen in the organs. In the present study, the mean values of the digital densitometric determination of collagen fibers identified by the first method did not differ significantly among the SB, PB and BB groups. Lung hydroxyproline content showed similar behavior to the picrosirius staining, suggesting equally that the DNA-hsp65 vaccine, in the present model, had no effect on the degree of bleomycin-induced collagen deposition.
Since no significant differences were detected in the measurements that reflect the degree of collagen deposition, the alterations in septal thickness detected in the H&E stainings are likely explained by effects on other histological elements. The interstitial septum is composed of cellular and extracellular components. In the subjective histological evaluation of the slides by a pulmonary pathologist, the SS group presented no abnormalities, although there was remodeling of the lung architecture in the SB and PB groups. Those last two groups presented mixed cell inflammatory infiltrate, predominantly lymphoplasmacytic, and areas of tissue repair located preferentially around the bronchovascular axis, extending into the adjacent parenchyma. In relation to the BB group, less intense injuries were observed, with new tissue repair, restricted to the central axis of the airways. Therefore, the effects of the DNA-hsp65 vaccine seem to have been more evident in the reduction of the intensity of the mixed cell inflammatory infiltrates than in the process of collagen deposition per se. It is equally relevant that, in addition to collagen, the extracellular matrix is composed of other biochemical elements, such as elastin, proteoglycans, laminin and fibronectin. Therefore, the protective effects of the vaccine might have occurred in some or all these last components.(26)
Recent advances in the understanding of the pathogenesis of IPF call into question the role of inflammatory alterations as primordial factors for the establishment of pulmonary fibrosis and suggest that epithelial elements associated with abnormal cicatrization processes are major primordial elements of the disease.(27) In the animal model of bleomycin-induced pulmonary fibrosis, however, previous studies indicate the existence of a correlation between the degree of the acute inflammatory alterations and later establishment of fibrosis.(28) In the present study, all animals were studied at the same time, that is, eight weeks after the end of immunization with the vaccine and six weeks after the IT instillation of bleomycin. We cannot exclude the possibility that sacrificing the animals at a later date, with greater evolution of the fibrotic process, might produce different results, particularly regarding a greater influence of immunization on the establishment of the fibrotic injuries.
We highlight that this is only a preliminary experimental study, and that other schedules with higher doses of the DNA-hsp65 vaccine or shorter intervals of administration might, in theory, provide better results. In addition, indeed, there are no animal models that fully reproduce the usual interstitial pneumonia-type injuries, which are the anatomopathologicalsubstrates of IPF. Therefore, the true role of the DNA-hsp65 vaccine in the treatment of pulmonary fibrosis, particularly IPF, in theory, would only be truly determined through the use of well-designed clinical assays.
In summary, we can conclude that the data here obtained indicate that immunization with the DNA-hsp65 vaccine had a positive effect on the degree of deposition of the noncollagen matrix in a model of bleomycin-induced lung injury. Additional studies are necessary in order to elucidate more fully the effects that the vaccine has on pulmonary fibrotic processes.
References 1. American Thoracic Society; European Respiratory Society. American Thoracic Society/European Respiratory Society International Multidisciplinary Consensus Classification of the Idiopathic Interstitial Pneumonias. This joint statement of the American Thoracic Society (ATS), and the European Respiratory Society (ERS) was adopted by the ATS board of directors, June 2001 and by the ERS Executive Committee, June 2001. Am J Respir Crit Care Med. 2002;165(2):277-304. Review. No abstract available. Erratum in: Am J Respir Crit Care Med. 2002;166(3):426.
2. American Thoracic Society. Idiopathic pulmonary fibrosis: diagnosis and treatment. International consensus statement. American Thoracic Society (ATS), and the European Respiratory Society (ERS). Am J Respir Crit Care Med. 2000;161(2 Pt 1):646-64.
3. Nathan SD. Therapeutic management of idiopathic pulmonary fibrosis: an evidence-based approach. Clin Chest Med. 2006;27(1 Suppl 1):S27-35, vi.
4. Haslam PL. Evaluation of alveolitis by studies of lung biopsies. Lung. 1990;168 Suppl:984-92.
5. Daniil Z, Kitsanta P, Kapotsis G, Mathioudaki M, Kollintza A, Karatza M, et al. CD8+ T lymphocytes in lung tissue from patients with idiopathic pulmonary fibrosis. Respir Res. 2005;6:81.
6. Gross TJ, Hunninghake GW. Idiopathic pulmonary fibrosis. N Engl J Med. 2001;345(7):517-25.
7. Singh S, du Bois R. Autoantibodies in cryptogenic fibrosing alveolitis. Respir Res. 2001;2(2):61-3.
8. Wallace WA, Ramage EA, Lamb D, Howie SE. A type 2 (Th2-like) pattern of immune response predominates in the pulmonary interstitium of patients with cryptogenic fibrosing alveolitis (CFA). Clin Exp Immunol. 1995;101(3):436-41.
9. Martinez JA, King TE Jr, Brown K, Jennings CA, Borish L, Mortenson RL, et al. Increased expression of the interleukin-10 gene by alveolar macrophages in interstitial lung disease. Am J Physiol. 1997;273(3 Pt 1):676-83.
10. Hancock A, Armstrong L, Gama R, Millar A. Production of interleukin 13 by alveolar macrophages from normal and fibrotic lung. Am J Respir Cell Mol Biol. 1998;18(1):60-5.
11. Lowrie DB, Tascon RE, Colston MJ, Silva CL. Towards a DNA vaccine against tuberculosis. Vaccine. 1994;12(16):1537-40.
12. Tascon RE, Colston MJ, Ragno S, Stavropoulos E, Gregory D, Lowrie DB. Vaccination against tuberculosis by DNA injection. Nat Med. 1996;2(8):888-92.
13. Lowrie DB, Silva CL, Tascon RE. DNA vaccines against tuberculosis. Immunol Cell Biol. 1997;75(6):591-4.
14. Bonato VL, Lima VM, Tascon RE, Lowrie DB, Silva CL. Identification and characterization of protective T cells in hsp65 DNA-vaccinated and Mycobacterium tuberculosis-infected mice. Infect Immun. 1998;66(1):169-75.
15. Lowrie DB, Tascon RE, Bonato VL, Lima VM, Faccioli LH, Stavropoulos E, et al. Therapy of tuberculosis in mice by DNA vaccination. Nature. 1999;400(6741):269-71.
16. Junqueira LC, Bignolas G, Brentani RR. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue sections. Histochem J. 1979;11(4):447-55.
17. Frankel SK, Moats-Staats BM, Cool CD, Wynes MW, Stiles AD, Riches DW. Human insulin-like growth factor-IA expression in transgenic mice promotes adenomatous hyperplasia but not pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol. 2005;288(5):805-12.
18. Walter N, Collard HR, King TE Jr. Current perspectives on the treatment of idiopathic pulmonary fibrosis. Proc Am Thorac Soc. 2006;3(4):330-8.
19. Yildirim Z, Kotuk M, Iraz M, Kuku I, Ulu R, Armutcu F, et al. Attenuation of bleomycin-induced lung fibrosis by oral sulfhydryl containing antioxidants in rats: erdosteine and N-acetylcysteine. Pulm Pharmacol Ther. 2005;18(5):367-73.
20. Martinez JA, Ramos SG, Meirelles MS, Verceze AV, Arantes MR, Vannucchi H. Effects of quercetin on bleomycin-induced lung injury: a preliminary study. J Bras Pneumol. 2008;34(7):445-52.
21. Silva CL, Bonato VL, Coelho-Castelo AA, De Souza AO, Santos SA, Lima KM, et al. Immunotherapy with plasmid DNA encoding mycobacterial hsp65 in association with chemotherapy is a more rapid and efficient form of treatment for tuberculosis in mice. Gene Ther. 2005;12(3):281-7.
22. Lima KM, Bonato VL, Faccioli LH, Brandão IT, Santos SA, Coelho-Castelo AA et al. Comparison of different delivery systems of vaccination for the induction of protection against tuberculosis in mice. Vaccine. 2001;19(25-26):3518-25.
23. Smith RB. Avaliação da eficácia da injeção da vacina de DNA HSP65 do Mycobacterium leprae em pacientes com estádio avançado de carcinoma epidermóide de cabeça e pescoço, em ensaio clínico de fase I/II. [thesis]. São Paulo: Universidade de São Paulo; 2007.
24. Gurujeyalakshmi G, Giri SN. Molecular mechanisms of antifibrotic effect of interferon gamma in bleomycin-mouse model of lung fibrosis: downregulation of TGF-beta and procollagen I and III gene expression. Exp Lung Res. 1995;21(5):791-808.
25. Raghu G, Brown KK, Bradford WZ, Starko K, Noble PW, Schwartz DA, et al. A placebo-controlled trial of interferon gamma-1b in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2004;350(2):125-33.
26. Roman J. Extracellular matrix in the pathogenesis of lung injury and repair. In: Schwarz M, King TE, editors. Interstitial lung disease. Hamilton: BC Decker; 2003. p. 277-99.
27. Selman M, King TE, Pardo A; American Thoracic Society; European Respiratory Society; American College of Chest Physicians. Idiopathic pulmonary fibrosis: prevailing and evolving hypotheses about its pathogenesis and implications for therapy. Ann Intern Med. 2001;134(2):136-51.
28. Shen AS, Haslett C, Feldsien DC, Henson PM, Cherniack RM. The intensity of chronic lung inflammation and fibrosis after bleomycin is directly related to the severity of acute injury. Am Rev Respir Dis. 1988;137(3):564-71.
Study conducted at the University of São Paulo at Ribeirão Preto School of Medicine, Ribeirão Preto, Brazil.
1. Attending Physician in the Pulmonology Division of the Department of Clinical Medicine. University of São Paulo at Ribeirão Preto School of Medicine, Ribeirão Preto, Brazil.
2. Full Professor in the Department of Biochemistry and Immunology. University of São Paulo at Ribeirão Preto School of Medicine, Ribeirão Preto, Brazil.
3. Professor in the Department of Pathology. University of São Paulo at Ribeirão Preto School of Medicine, Ribeirão Preto, Brazil.
4. Full Professor in the Department of Clinical, Toxicological and Bromatological Analysis. University of São Paulo at Ribeirão Preto School of Medicine, Ribeirão Preto, Brazil.
5. Associate Professor in the Pulmonology Division of the Department of Clinical Medicine. University of São Paulo at Ribeirão Preto School of Medicine, Ribeirão Preto, Brazil.
Correspondence to: José Antônio Baddini Martinez. Departamento de Clínica Médica, Avenida Bandeirantes, 3900, Campus Universitário, CEP 14048-900, Ribeirão Preto, SP, Brazil.
Tel 55 16 602-2531. E-mail: jabmarti@fmrp.usp.br
Financial Support: This study received financial support from the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Foundation for the Support of Research in the State of São Paulo).
Submitted: 24 January 2008. Accepted, after review: 25 February 2008.






